

Earth Science Frontiers ›› 2021, Vol. 28 ›› Issue (5): 104-113.DOI: 10.13745/j.esf.sf.2021.2.20
Previous Articles Next Articles
DUAN Lei( ), YANG Shengke, WANG Wenke*(
), YANG Shengke, WANG Wenke*( )
)
Received:2020-05-15
Revised:2020-08-20
Online:2021-09-25
Published:2021-10-29
Contact:
WANG Wenke
CLC Number:
DUAN Lei, YANG Shengke, WANG Wenke. Effect of humic acid on the phase distribution of sulfadiazine in shallow groundwater[J]. Earth Science Frontiers, 2021, 28(5): 104-113.
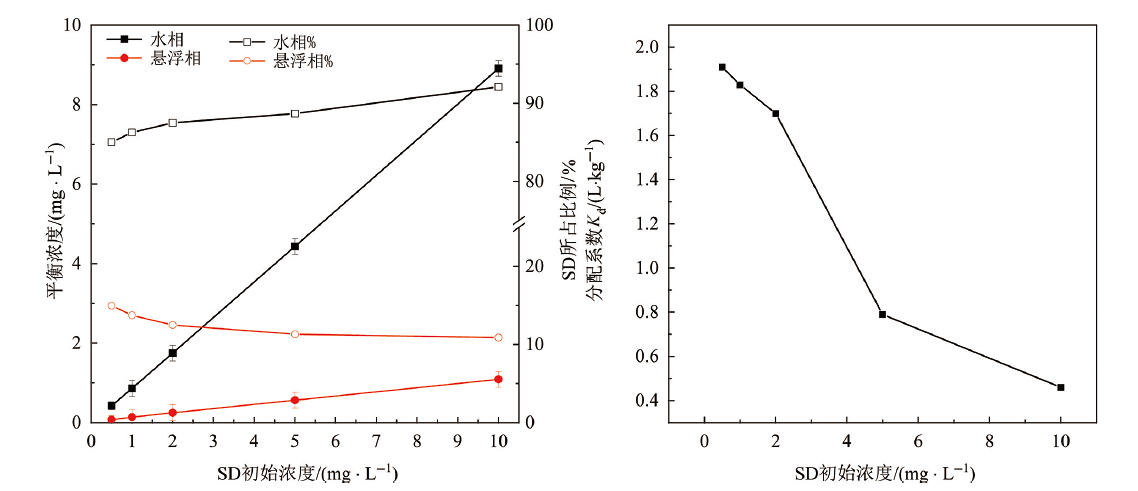
Fig.3 Sulfadiazine partition between phases at equilibrium (left) and SD phase distribution coefficient Kd(right) as a function of SD initial concentration
| 抗生素 | pH 值 | Ksv/ (103L·mol-1) | Kq/ (1011L·mol-1·s-1) | Kb/ (103L·mol-1) | n |
|---|---|---|---|---|---|
| SD | 4 | 3.894 | 3.894 | 3.724 | 1.34 |
| 8 | 4.043 | 4.043 | 4.015 | 0.69 | |
| 10 | 3.946 | 3.946 | 3.824 | 0.81 |
Table 1 Thermodynamic parameters for SD-HA interaction at different temperatures
| 抗生素 | pH 值 | Ksv/ (103L·mol-1) | Kq/ (1011L·mol-1·s-1) | Kb/ (103L·mol-1) | n |
|---|---|---|---|---|---|
| SD | 4 | 3.894 | 3.894 | 3.724 | 1.34 |
| 8 | 4.043 | 4.043 | 4.015 | 0.69 | |
| 10 | 3.946 | 3.946 | 3.824 | 0.81 |
| [1] |
GUO X, YIN Y, YANG C, et al. Remove mechanisms of sulfamethazine by goethite: the contributions of pH and ionic strength[J]. Research on Chemical Intermediates, 2016, 42(7):6423-6435.
DOI URL |
| [2] | FAN Z, CASEY F X M, HAKK H , et al. Sorption, fate, and mobility of sulfonamides in soils[J]. Water Air & Soil Pollution, 2011, 218(1/2/3/4):49-61. |
| [3] |
ISLAS-ESPINOZA M, REID B J, WEXLER M, et al. Soil bacterial consortia and previous exposure enhance the biodegradation of sulfonamides from pig manure[J]. Microbial Ecology, 2012, 64(1):140-151.
DOI URL |
| [4] | 金磊, 姜蕾, 韩琪, 等. 华东地区某水源水中13种磺胺类抗生素的分布特征及人体健康风险评价[J]. 环境科学, 2016, 37(7):2515-2521. |
| [5] | 金彩霞, 司晓薇, 王子英, 等. 养殖场周边土壤-蔬菜系统磺胺类药物残留及风险评价[J]. 环境科学, 2016, 37(4):1562-1567. |
| [6] |
BARNES K K, KOLPIN D W, FURLONG E T, et al. A national reconnaissance of pharmaceuticals and other organic wastewater contaminanants in the United States-Ⅰ groundwater[J]. Science of the Total Environment, 2008, 402(2/3):192-200.
DOI URL |
| [7] |
JURADO A, CZRRERA J, PETROVIC M, et al. Occurrence of 95 pharmaceuticals and transformation products in urban groundwaters underlying the metropolis of Barcelona, Spain[J]. Environmental Pollution, 2013, 174:305-315.
DOI URL |
| [8] |
SIBLEY S D, PEDERSEN J A. Interaction of the macrolide antimicrobial clarithromycin with dissolved humic acid[J]. Environmental Science & Technology, 2008, 42(2):422-428.
DOI URL |
| [9] |
THIELE-BRUHN S, SEIBICKE T, SCHULTEN H R, et al. Sorption of sulfonamide pharmaceutical antibiotics on whole soils and particle-size fractions[J]. Journal of Environmental Quality, 2004, 33(4):1331-1342.
DOI URL |
| [10] |
ZHOU P, YAN H, GU B. Competitive complexation of metal ions with humic substances[J]. Chemosphere, 2005, 58(10):1327-1337.
DOI URL |
| [11] |
BLACKWELL P A, KAY P, ASHAUER R, et al. Effects of agricultural conditions on the leaching behaviour of veterinary antibiotics in soils[J]. Chemosphere, 2009, 75(1):13-19.
DOI URL |
| [12] |
CARMOSINI N, LEE L S. Ciprofloxacin sorption by dissolved organic carbon from reference and bio-waste materials[J]. Chemosphere, 2009, 77(6):813-820.
DOI URL |
| [13] | 朱先辰. 淡水环境中溶解性有机物及其与Cu2+相互作用: 荧光光谱学研究[D]. 武汉: 华中科技大学, 2014. |
| [14] | 周雪. 光谱学技术探测磺胺类药物与人血清白蛋白的相互作用[D]. 南昌: 南昌大学, 2016. |
| [15] | GUO Y, QIN P, WANG C, et al. Characterization on the toxic mechanism of two fluoroquinolones to trypsin by spectroscopic and computational methods[J]. Journal of Environmental Science and Health Part B (Pesticides Food Contaminants and Agricultural Wastes), 2019, 55(3):230-238. |
| [16] | 李仲谨, 李铭杰, 王海峰, 等. 腐殖酸类物质应用研究进展[J]. 化学研究, 2009, 20(4):103-107. |
| [17] | 陈景元, 王果庭, 刘金柱. 稳态荧光猝灭法确定胶束聚集数的研究[J]. 物理化学学报, 1993, 9(4):461-465. |
| [18] | MARTíNEZ L, BILSKI P, CHIGNELL C F. Effect of magnesium and calcium complexation on the photochemical properties of Norfloxacin[J]. Photochemistry & Photobiology, 1996, 64(6):911-917. |
| [19] | 李思静. 胡敏酸胶体对DDT在河床沉积物中吸附与运移的影响[D]. 西安: 长安大学, 2012. |
| [20] |
PAVEL J, VACLAV H, PETRA B, et al. Reduction and immobilization of hexavalent chromium with coal- and humate-based sorbents[J]. Chemosphere, 2009, 75(6):732-738.
DOI URL |
| [21] | 王俊. 腐殖酸对砷在土壤中的形态转化和生物有效性的影响研究[D]. 重庆: 西南大学, 2017. |
| [22] | 刘阳洋. 腐殖酸对海拉尔河典型抗生素污染物吸附作用研究[D]. 西安: 西安科技大学, 2017. |
| [23] | 李璐, 王震宇, 林道辉, 等. 天然有机质与重金属相互作用的分析方法进展[J]. 环境科学研究, 2015, 28(2):182-189. |
| [24] | 张红梅, 王彦卿, 周秋华, 等. 腐殖酸与吡虫啉作用的荧光光谱研究[J]. 化学试剂, 2009, 31(9):679-681. |
| [25] | 钱正英, 张光斗. 中国可持续发展水资源战略研究综合报告及各专题报告[R]. 北京: 中国水利水电出版社, 2001. |
| [26] | 孙梅香, 刘文, 高嘉苓, 等. 荧光光谱法研究草甘膦与腐殖酸的相互作用[J]. 环境化学, 2015, 34(8):1529-1534. |
| [27] | WANG R, YANG S, FANG J, et al. Characterizing the interaction between antibiotics and humic acid by fluorescence quenching method[J]. International Journal of Environmental Research & Public Health, 2018, 15(7):1458. |
| [28] | XIAOYU Y, SHENGKE Y, JIE F, et al. Interaction mechanism between antibiotics and humic acid by UV-Vis spectrometry[J]. International Journal of Environmental Research & Public Health, 2018, 15(9):1911. |
| [29] | DANTAS M D D A, SILVA M D M, SILVA O N, et al. Interactions of tetracyclines with milk allergenic protein (casein): a molecular and biological approach[J]. Journal of Biomolecular Structure & Dynamics, 2019, 38(18):1-21. |
| [30] |
GUO H, JIANG N, WANG H, et al. Enhanced catalytic performance of graphene-TiO2 nanocomposites for synergetic degradation of fluoroquinolone antibiotic in pulsed discharge plasma system[J]. Applied Catalysis B: Environmental, 2019, 248(5):552-566.
DOI URL |
| [31] | 于泳. 多环芳烃和壬基酚及前体物在水-沉积物界面分配行为研究[D]. 天津: 南开大学, 2009. |
| [32] | 傅平青, 刘丛强, 尹祚莹, 等. 腐殖酸三维荧光光谱特性研究[J]. 地球化学, 2004, 33(3):301-308. |
| [33] | 方洁. 抗生素在水体中相态间分布规律及与胡敏酸作用机制研究[D]. 西安: 长安大学, 2018. |
| [34] | 张步迪, 林青, 曹东平, 等. 磺胺嘧啶在土壤及土壤组分中的吸附/解吸动力学[J]. 土壤, 2018, 50(5):949-957. |
| [35] | 兰亚琼. 水环境中镉离子与腐殖酸作用特性的研究[D]. 西安: 西安建筑科技大学, 2011. |
| [36] | 朱江鹏, 梅婷, 彭云, 等. 荧光猝灭法研究洛克沙胂与腐殖酸的相互作用[J]. 环境科学, 2014, 35(7):2620-2626. |
| [37] |
XU H, YU G, YANG L, et al. Combination of two-dimensional correlation spectroscopy and parallel factor analysis to characterize the binding of heavy metals with DOM in lake sediments[J]. Journal of Hazardous Materials, 2013, 263(2):412-421.
DOI URL |
| [38] | 韩甜甜. 水体中共存污染物与抗生素相互作用的荧光光谱法研究[D]. 宜昌: 三峡大学, 2012. |
| [39] |
GORDON E, BROWN J, GEORGE A. Sorption of trace elements on mineral surfaces: modern perspectives from spectroscopic studies, and comments on sorption in the marine environment[J]. International Geology Review, 2001, 43(11):963-1073.
DOI URL |
| [40] | 孙绍发, 刘洁, 宋功武. 荧光光谱法研究铁(Ⅲ)存在下依诺沙星与牛血清白蛋白的相互作用[J]. 分析科学学报, 2007, 23(2):166-168. |
| [41] | 张勇庞, 包维楷. 土壤有机质及其研究方法综述[J]. 世界科技研究与发展, 2005, 27(5):72-78. |
| [42] | 王志军, 吴群, 雷海英. 柠檬酸钠与牛血清白蛋白相互作用的荧光光谱研究[J]. 光谱实验室, 2012, 29(2):847-851. |
| [43] | 于兵川, 吴洪特, 周培疆, 等. 五氯苯酚与腐殖酸作用的荧光猝灭效应研究[J]. 环境化学, 2006, 25(2):164-167. |
| [44] |
GAO J, PEDERSEN J A. Adsorption of sulfonamide antimicrobial agents to clay minerals[J]. Environmental Science & Technology, 2005, 39(24):9509-9516.
DOI URL |
| [45] |
TOLLS J. Sorption of veterinary pharmaceuticals in soils: a review[J]. Environmental Science & Technology, 2001, 35(17):3397-3406.
DOI URL |
| [46] | 李毓骐, 朱亚先, 鹿贞彬, 等. 荧光极性探针在腐殖酸和芘相互作用研究中的应用[J]. 厦门大学学报(自然科学版), 2007, 46(1):59-62. |
| [47] | 李文慧, 李爱民, 王学军. pH值和离子强度对胡敏酸与芘之间相互作用的影响[J]. 环境化学, 2009, 28(5):636-639. |
| [48] | 马连刚, 肖保华, 唐婷. 胡敏酸衰减全反射-傅里叶变换-红外光谱研究[J]. 地球与环境, 2014, 42(1):55-61. |
| [1] | CHEN Hongwei, YANG Yao, HUANG He, ZHOU Hui, PENG Xiangxun, YU Shasha, YU Weihou, LI Zhengzui, WANG Zhaoguo. Interaction between surface water and groundwater during the dry season in Lake Dongting based on 222Rn tracing [J]. Earth Science Frontiers, 2024, 31(2): 423-434. |
| [2] | ZHANG Yanbin, ZHAI Mingguo, ZHOU Yanyan, ZHOU Ligang. The continental lower crust [J]. Earth Science Frontiers, 2024, 31(1): 28-45. |
| [3] | JIN Zhijun, CHEN Shuping, ZHANG Rui. Fluctuation analysis for sedimentary basins: Review and outlook [J]. Earth Science Frontiers, 2024, 31(1): 284-296. |
| [4] | DONG Hailiang, ZENG Qiang, LIU Deng, SHENG Yizhi, LIU Xiaolei, LIU Yuan, HU Jinglong, LI Yang, XIA Qingyin, LI Runjie, HU Dafu, ZHANG Donglei, ZHANG Wenhui, GUO Dongyi, ZHANG Xiaowen. Interactions between clay minerals and microbes: Mechanisms and applications in environmental remediation [J]. Earth Science Frontiers, 2024, 31(1): 467-485. |
| [5] | LAN Chunyuan, ZHANG Lifei, TAO Renbiao, HU Han, ZHANG Lijuan, WANG Chao. Calculation methods for fluid composition and water-rock interaction in the deep Earth based on DEW model—a review [J]. Earth Science Frontiers, 2024, 31(1): 64-76. |
| [6] | ZHANG Guanglu, LIU Haiyan, GUO Huaming, SUN Zhanxue, WANG Zhen, WU Tonghang. Occurrences and health risks of high-nitrate groundwater in typical piedmont areas of the North China Plain [J]. Earth Science Frontiers, 2023, 30(4): 485-503. |
| [7] | WANG Zhen, GUO Huaming, LIU Haiyan, XING Shiping. Geochemical characteristics of rare earth elements in high-fluoride groundwater in the Guide Basin and its implications [J]. Earth Science Frontiers, 2023, 30(3): 505-514. |
| [8] | GUO Xiaoyu, LUO Xucong, GAO Rui, XU Xiao, LU Zhanwu, HUANG Xingfu, LI Wenhui, LI Chunsen. Crustal-scale plate interactions beneath the dominant domain in the India-Eurasia collision zone—a tectonogeophysical study [J]. Earth Science Frontiers, 2023, 30(2): 1-17. |
| [9] | XING Shiping, WU Ping, HU Xueda, GUO Huaming, ZHAO Zhen, YUAN Youjing. Geochemical characteristics of aquifer sediments and their influence on fluoride enrichment in groundwater in the Hualong-Xunhua basin [J]. Earth Science Frontiers, 2023, 30(2): 526-538. |
| [10] | WU Lixin, JING Zhao, CHEN Xianyao, LI Caiwen, ZHANG Guoliang, WANG Shi, DONG Bo, ZHUANG Guangchao. Marine science in China: Current status and future outlooks [J]. Earth Science Frontiers, 2022, 29(5): 1-12. |
| [11] | ZHANG Xu, ZHANG Weichen, WANG Zhenqian, ZHENG Kai, DENG Fengfei. The Glacial North Atlantic “Capacitor” effect controlled by seasonal sea ice change [J]. Earth Science Frontiers, 2022, 29(5): 334-341. |
| [12] | WANG Guangcai, WANG Yanxin, LIU Fei, GUO Huaming. Advances and trends in hydrogeochemical studies: Insights from bibliometric analysis [J]. Earth Science Frontiers, 2022, 29(3): 25-36. |
| [13] | LIU Haiyan, LIU Maohan, ZHANG Weimin, SUN Zhanxue, WANG Zhen, WU Tonghang, GUO Huaming. Distribution and fractionation of rare earth elements in high fluoride groundwater from the North China Plain [J]. Earth Science Frontiers, 2022, 29(3): 129-144. |
| [14] | XING Shiping, GUO Huaming, WU Ping, HU Xueda, ZHAO Zhen, YUAN Youjing. Distribution and formation processes of high fluoride groundwater in different types of aquifers in the Hualong-Xunhua Basin [J]. Earth Science Frontiers, 2022, 29(3): 115-128. |
| [15] | GUO Huaming, GAO Zhipeng, XIU Wei. Typical redox-sensitive components in groundwater systems: Research highlights and trends [J]. Earth Science Frontiers, 2022, 29(3): 64-75. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||