

地学前缘 ›› 2022, Vol. 29 ›› Issue (3): 217-226.DOI: 10.13745/j.esf.sf.2022.1.30
崔迪1( ), 杨冰2, 郭华明1,*(
), 杨冰2, 郭华明1,*( ), 连国玺2, 孙娟2
), 连国玺2, 孙娟2
收稿日期:2021-12-02
修回日期:2022-01-21
出版日期:2022-05-25
发布日期:2022-04-28
通讯作者:
郭华明
作者简介:崔 迪(1996—),女,硕士研究生,水文地质学专业。E-mail: 15972112240@163.com
基金资助:
CUI Di1( ), YANG Bing2, GUO Huaming1,*(
), YANG Bing2, GUO Huaming1,*( ), LIAN Guoxi2, SUN Juan2
), LIAN Guoxi2, SUN Juan2
Received:2021-12-02
Revised:2022-01-21
Online:2022-05-25
Published:2022-04-28
Contact:
GUO Huaming
摘要:
地浸铀矿山退役后,含水层中残留的含铀浸出液随着地下水的运动向下游迁移扩散,存在对周边地下水污染的风险。本文设计了若干组批实验和柱实验,研究铀在北方某地浸铀矿山砂岩含水介质中的吸附和迁移行为。实验结果表明,砂岩对铀的吸附在12 h以内达到平衡,铀初始浓度越高,砂岩的铀吸附容量越大;砂岩对铀的吸附为吸热反应,温度升高有利于吸附反应的进行。溶液pH值和共存
中图分类号:
崔迪, 杨冰, 郭华明, 连国玺, 孙娟. 砂岩含水介质中铀的吸附和迁移行为研究[J]. 地学前缘, 2022, 29(3): 217-226.
CUI Di, YANG Bing, GUO Huaming, LIAN Guoxi, SUN Juan. Adsorption and transport of uranium in porous sandstone media[J]. Earth Science Frontiers, 2022, 29(3): 217-226.
| 柱编号 | pH值 | 铀浓度/ (mg·L-1) | 流速/ (mL·min-1) | (mmol·L-1) | |
|---|---|---|---|---|---|
| 1 | 4 | 2 | 0.1 | 0 | |
| 2 | 7 | 2 | 0.1 | 0 | |
| 3 | 8.5 | 2 | 0.1 | 0 | |
| 4 | 7 | 1 | 0.1 | 0 | |
| 5 | 7 | 5 | 0.1 | 0 | |
| 6 | 7 | 2 | 0.5 | 0 | |
| 7 | 7 | 2 | 0.3 | 0 | |
| 8 | 7 | 2 | 0.1 | 0.5 | |
| 9 | 7 | 2 | 0.1 | 1.0 |
表1 柱实验条件
Table 1 Experimental conditions in column experiments
| 柱编号 | pH值 | 铀浓度/ (mg·L-1) | 流速/ (mL·min-1) | (mmol·L-1) | |
|---|---|---|---|---|---|
| 1 | 4 | 2 | 0.1 | 0 | |
| 2 | 7 | 2 | 0.1 | 0 | |
| 3 | 8.5 | 2 | 0.1 | 0 | |
| 4 | 7 | 1 | 0.1 | 0 | |
| 5 | 7 | 5 | 0.1 | 0 | |
| 6 | 7 | 2 | 0.5 | 0 | |
| 7 | 7 | 2 | 0.3 | 0 | |
| 8 | 7 | 2 | 0.1 | 0.5 | |
| 9 | 7 | 2 | 0.1 | 1.0 |
| 柱号 | 饱和含 水率θ | 流速 v/(cm·min-1) | 弥散系数 D/(cm2·min-1) | 弥散度 λ/cm | R2 |
|---|---|---|---|---|---|
| 1 | 0.333 | 0.309 | 0.011 | 0.034 | 0.998 |
| 2 | 0.411 | 0.308 | 0.012 | 0.037 | 0.998 |
| 3 | 0.457 | 0.270 | 0.009 | 0.034 | 0.993 |
| 4 | 0.342 | 0.330 | 0.028 | 0.085 | 0.982 |
| 5 | 0.346 | 0.342 | 0.018 | 0.053 | 0.995 |
| 6 | 0.420 | 0.959 | 0.087 | 0.091 | 0.999 |
| 7 | 0.389 | 0.585 | 0.011 | 0.019 | 0.993 |
| 8 | 0.405 | 0.297 | 0.021 | 0.072 | 0.985 |
| 9 | 0.447 | 0.287 | 0.018 | 0.064 | 0.999 |
表2 水力学特征参数
Table 2 Characteristic hydraulic parameters
| 柱号 | 饱和含 水率θ | 流速 v/(cm·min-1) | 弥散系数 D/(cm2·min-1) | 弥散度 λ/cm | R2 |
|---|---|---|---|---|---|
| 1 | 0.333 | 0.309 | 0.011 | 0.034 | 0.998 |
| 2 | 0.411 | 0.308 | 0.012 | 0.037 | 0.998 |
| 3 | 0.457 | 0.270 | 0.009 | 0.034 | 0.993 |
| 4 | 0.342 | 0.330 | 0.028 | 0.085 | 0.982 |
| 5 | 0.346 | 0.342 | 0.018 | 0.053 | 0.995 |
| 6 | 0.420 | 0.959 | 0.087 | 0.091 | 0.999 |
| 7 | 0.389 | 0.585 | 0.011 | 0.019 | 0.993 |
| 8 | 0.405 | 0.297 | 0.021 | 0.072 | 0.985 |
| 9 | 0.447 | 0.287 | 0.018 | 0.064 | 0.999 |
| U初始浓度/ (mg·L-1) | 实验q'e/ (102 mg·g-1) | 准一级动力学 | 准二级动力学 | |||
|---|---|---|---|---|---|---|
| R2 | qe/(102 mg·g-1) | k2/(g·mg-1·h-1) | R2 | |||
| 1 | 2.763 | 0.237 | 2.794 | 69.973 | 0.997 | |
| 2 | 5.268 | 0.443 | 5.357 | 24.928 | 0.993 | |
| 5 | 22.555 | 0.338 | 22.367 | 12.562 | 0.999 | |
表3 准一级和准二级动力学模型拟合参数表
Table 3 Parameters of psudo first order and psudo second order kinetic models for uranium adsorption on sandstone
| U初始浓度/ (mg·L-1) | 实验q'e/ (102 mg·g-1) | 准一级动力学 | 准二级动力学 | |||
|---|---|---|---|---|---|---|
| R2 | qe/(102 mg·g-1) | k2/(g·mg-1·h-1) | R2 | |||
| 1 | 2.763 | 0.237 | 2.794 | 69.973 | 0.997 | |
| 2 | 5.268 | 0.443 | 5.357 | 24.928 | 0.993 | |
| 5 | 22.555 | 0.338 | 22.367 | 12.562 | 0.999 | |
| 温度/℃ | Langmuir等温吸附方程 | Freundlich等温吸附方程 | |||||
|---|---|---|---|---|---|---|---|
| KL | qmax | R2 | KF | 1/n | R2 | ||
| 10 | 4.309 | 0.088 | 0.990 | 0.076 | 0.582 | 0.979 | |
| 25 | 7.276 | 0.070 | 0.980 | 0.076 | 0.553 | 0.994 | |
| 35 | 10.263 | 0.085 | 0.992 | 0.088 | 0.500 | 0.978 | |
| 45 | 30.325 | 0.105 | 0.994 | 0.149 | 0.453 | 0.969 | |
表4 Langmuir和Freundlich等温吸附模型拟合参数
Table 4 Langmuir and Freundlich constants for uranium adsorption on sandstone at different temperatures
| 温度/℃ | Langmuir等温吸附方程 | Freundlich等温吸附方程 | |||||
|---|---|---|---|---|---|---|---|
| KL | qmax | R2 | KF | 1/n | R2 | ||
| 10 | 4.309 | 0.088 | 0.990 | 0.076 | 0.582 | 0.979 | |
| 25 | 7.276 | 0.070 | 0.980 | 0.076 | 0.553 | 0.994 | |
| 35 | 10.263 | 0.085 | 0.992 | 0.088 | 0.500 | 0.978 | |
| 45 | 30.325 | 0.105 | 0.994 | 0.149 | 0.453 | 0.969 | |
| T/℃ | KL | ΔG0/ (kJ·mol-1) | ΔH0/ (kJ·mol-1) | ΔS0/ (J·mol-1·K-1) |
|---|---|---|---|---|
| 10 | 4.31 | -3.44 | 7.10 | 37.23 |
| 25 | 7.28 | -4.92 | 40.32 | |
| 35 | 10.26 | -5.96 | 42.40 | |
| 45 | 30.33 | -9.02 | 50.69 |
表5 热力学模型拟合参数
Table 5 Thermodynamic parameters for uranium adsorption on sandstone
| T/℃ | KL | ΔG0/ (kJ·mol-1) | ΔH0/ (kJ·mol-1) | ΔS0/ (J·mol-1·K-1) |
|---|---|---|---|---|
| 10 | 4.31 | -3.44 | 7.10 | 37.23 |
| 25 | 7.28 | -4.92 | 40.32 | |
| 35 | 10.26 | -5.96 | 42.40 | |
| 45 | 30.33 | -9.02 | 50.69 |
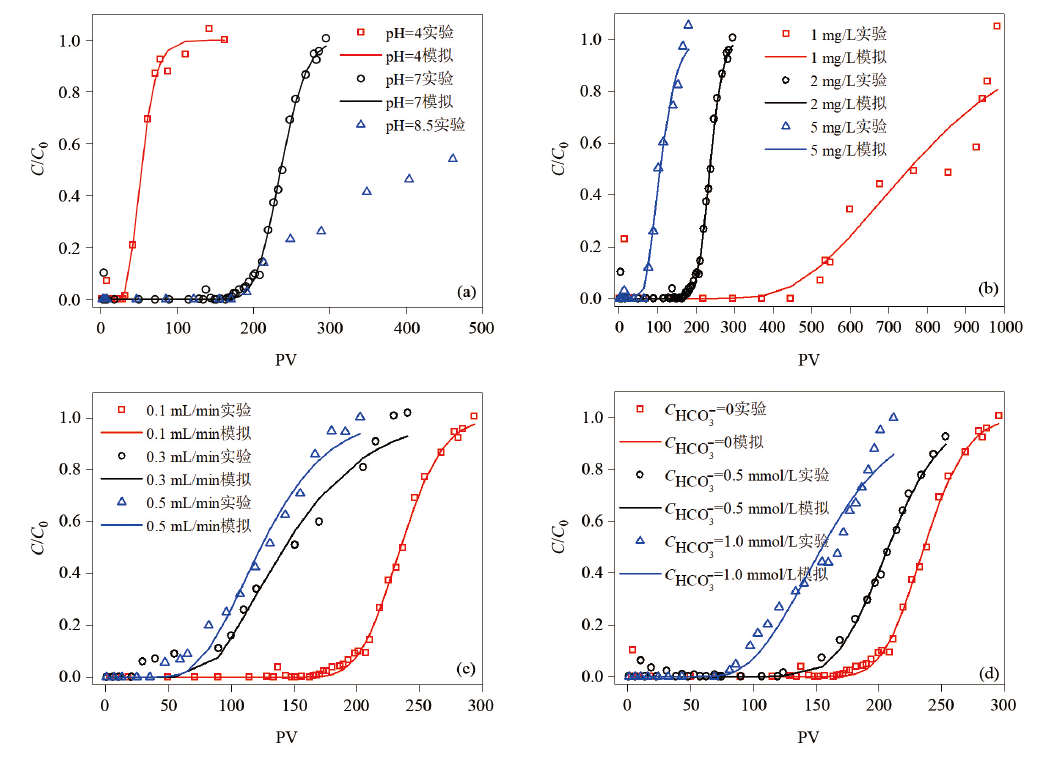
图4 不同条件下的铀穿透曲线
Fig.4 Breakthrough curves of uranium adsorption in sandstone columns under different conditions a—CU=2 mg/L, Q=0.1 mL/min, C H C O 3 -=0; b—pH=7, Q=0.1 mL/min, C H C O 3 -=0 mmol/L; c—pH=7, CU=2 mg/L, C H C O 3 -=0; and d—pH=7, CU=2 mg/L, Q=0.1 mL/min。
| 条件 | k'd/(mL·g-1) | ω/(103 min-1) | R2 |
|---|---|---|---|
| pH=4 | 9.82 | 1.720 | 0.995 |
| pH=7 | 117 | 0.182 | 0.997 |
| CU=1 mg/L | 159 | 0.122 | 0.936 |
| CU=2 mg/L | 117 | 0.182 | 0.997 |
| CU=5 mg/L | 23.9 | 0.828 | 0.989 |
| 流速0.1 mL/min | 117 | 0.182 | 0.974 |
| 流速0.3 mL/min | 49.5 | 0.604 | 0.977 |
| 流速0.5 mL/min | 39.5 | 1.228 | 0.997 |
| | 117 | 0.182 | 0.997 |
| | 58.9 | 0.483 | 0.996 |
| | 48.1 | 4.759 | 0.964 |
表6 模型拟合参数及拟合度系数
Table 6 The fitted parameters of breakthrough curves of uranium adsorption in sandstone column
| 条件 | k'd/(mL·g-1) | ω/(103 min-1) | R2 |
|---|---|---|---|
| pH=4 | 9.82 | 1.720 | 0.995 |
| pH=7 | 117 | 0.182 | 0.997 |
| CU=1 mg/L | 159 | 0.122 | 0.936 |
| CU=2 mg/L | 117 | 0.182 | 0.997 |
| CU=5 mg/L | 23.9 | 0.828 | 0.989 |
| 流速0.1 mL/min | 117 | 0.182 | 0.974 |
| 流速0.3 mL/min | 49.5 | 0.604 | 0.977 |
| 流速0.5 mL/min | 39.5 | 1.228 | 0.997 |
| | 117 | 0.182 | 0.997 |
| | 58.9 | 0.483 | 0.996 |
| | 48.1 | 4.759 | 0.964 |
| 条件 | 柱实验分配系数 k'd/(mL·g-1) | 批实验分配系数 kd/(mL·g-1) |
|---|---|---|
| pH=4 | 9.81 | 65.1 |
| pH=7 | 117 | 637 |
| CU=1 mg/L | 159 | 189 |
| CU=2 mg/L | 117 | 133 |
| CU=5 mg/L | 23.9 | 55.5 |
| | 117 | 133 |
| | 58.9 | 83.1 |
| | 48.1 | 69.0 |
表7 柱实验和批实验分配系数对比
Table 7 Partition coefficients from column and batch experiments
| 条件 | 柱实验分配系数 k'd/(mL·g-1) | 批实验分配系数 kd/(mL·g-1) |
|---|---|---|
| pH=4 | 9.81 | 65.1 |
| pH=7 | 117 | 637 |
| CU=1 mg/L | 159 | 189 |
| CU=2 mg/L | 117 | 133 |
| CU=5 mg/L | 23.9 | 55.5 |
| | 117 | 133 |
| | 58.9 | 83.1 |
| | 48.1 | 69.0 |
| [1] | 张晓峰, 陈迪云, 彭燕, 等. 丁二酸改性茶油树木屑吸附铀的研究[J]. 环境科学, 2015, 36(5): 1686-1693. |
| [2] | 黎广荣, 周义朋, 赵凯, 等. 砂岩型铀矿浸出矿物工艺学研究进展[J]. 有色金属(冶炼部分), 2021(8): 9-19. |
| [3] | 左维, 谭凯旋. 新疆某地浸采铀矿山退役井场地下水污染特征[J]. 南华大学学报(自然科学版), 2014, 28(4): 28-34. |
| [4] |
FU H Y, DING D X, SUI Y, et al. Transport of uranium(VI) in red soil in South China: influence of initial pH and carbonate concentration[J]. Environmental Science and Pollution Research, 2019, 26(36): 37125-37136.
DOI URL |
| [5] |
BRUGGEMAN C, MAES N. Uptake of uranium(VI) by pyrite under boom clay conditions: influence of dissolved organic carbon[J]. Environmental Science and Technology, 2010, 44(11): 4210-4216.
DOI URL |
| [6] | 廉欢, 高柏, 郭亚丹, 等. 某尾矿库区水环境中放射性核素铀的变化特征及影响因素[J]. 有色金属(冶炼部分), 2017(5): 64-68. |
| [7] |
ZHANG Z B, LIU J, CAO X H, et al. Comparison of U(VI) adsorption onto nanoscale zero-valent iron and red soil in the presence of U(VI)-CO3/Ca-U(VI)-CO3 complexes[J]. Journal of Hazardous Materials, 2015, 300: 633-642.
DOI URL |
| [8] |
GE M T, WANG D J, YANG J W, et al. Co-transport of U(VI) and akaganéite colloids in water-saturated porous media: role of U(VI) concentration, pH and ionic strength[J]. Water Research, 2018, 147: 350-361.
DOI URL |
| [9] | 惠淑君. 铀在砂岩含水层介质中的吸附和转化特征[D]. 北京: 中国地质大学(北京), 2020. |
| [10] | 惠淑君, 杨冰, 郭华明, 等. 不同因素对砂岩含水层介质吸附铀的影响[J]. 地学前缘, 2021, 28(5): 68-78. |
| [11] | 丛伟, 张兴昌, 封晔. 不同CDE模型对硒在黄绵土中运移特性的模拟研究[J]. 水土保持学报, 2011, 25(3): 220-224. |
| [12] |
MA J, MA Y L, WEI R F, et al. Phosphorus transport in different soil types and the contribution of control factors to phosphorus retardation[J]. Chemosphere, 2021, 276: 130012.
DOI URL |
| [13] | Šimůnek J, Šejna M, Saito H, et al. The HYDRUS-1D software package for simulating the one-dimensional movement of water, heat, and multiple solutes in variably-saturated media[R]. California: University of California Riverside, 2013. |
| [14] |
VAN GENUCHTEN M T, IMUNEK J Å, LEIJ F J, et al. STANMOD: model use, calibration, and validation[J]. Transactions of the ASABE. 2012, 55(4): 1355-1368.
DOI URL |
| [15] |
LI S C, WANG X L, HUANG Z Y, et al. Sorption and desorption of uranium(VI) on GMZ bentonite: effect of pH, ionic strength, foreign ions and humic substances[J]. Journal of Radioanalytical and Nuclear Chemistry, 2016, 308(3): 877-886.
DOI URL |
| [16] | 李松南. 以蛋壳为原料制备多种吸附材料及其铀吸附性能研究[D]. 哈尔滨: 哈尔滨工程大学, 2013. |
| [17] | 赵凯. 改性天然菱铁矿除砷性能与应用[D]. 北京: 中国地质大学(北京), 2014. |
| [18] | 赵凯, 郭华明, 李媛, 等. 天然菱铁矿改性及强化除砷研究[J]. 环境科学, 2012, 33(2): 459-468. |
| [19] |
GUO H M, LI Y, ZHAO K. Arsenate removal from aqueous solution using synthetic siderite[J]. Journal of Hazardous Materials, 2010, 176(1/2/3): 174-180.
DOI URL |
| [20] |
SHAN Y, GUO H M. Fluoride adsorption on modified natural siderite: optimization and performance[J]. Chemical Engineering Journal, 2013, 223: 183-191.
DOI URL |
| [21] | 扶海鹰. 红壤中铀的迁移转化规律及控制方法研究[D]. 衡阳: 南华大学, 2019. |
| [22] | 葛孟团. 胡敏酸和针铁矿胶体对U(Ⅵ)在饱和花岗岩颗粒柱中运移的影响研究[D]. 兰州: 兰州大学, 2019. |
| [23] |
BACHMAF S, PLANER-FRIEDRICH B, MERKEL B J. Effect of sulfate, carbonate, and phosphate on the uranium(VI) sorption behavior onto bentonite[J]. Radiochimica Acta, 2008, 96(6): 359-366.
DOI URL |
| [24] |
YANG Z W, KANG M L, MA B, et al. Inhibition of U(VI) reduction by synthetic and natural pyrite[J]. Environmental Science and Technology, 2014, 48(18): 10716-10724.
DOI URL |
| [25] | 杜作勇, 王彦惠, 李东瑞, 等. 膨润土对U(Ⅵ)的吸附机理研究[J]. 核技术, 2019, 42(2): 22-29. |
| [26] |
CUMBERLAND S A, WILSON S A, ETSCHMANN B, et al. Rapid immobilisation of U(VI) by eucalyptus bark: adsorption without reduction[J]. Applied Geochemistry, 2018, 96: 1-10.
DOI URL |
| [27] | 葛孟团. 水合氧化铁胶体对U(Ⅵ)在饱和石英砂柱中运移的影响[D]. 兰州: 兰州大学, 2016. |
| [28] | 白静, 赵勇胜, 秦传玉. 重金属在含水层迁移实验方案设计[J]. 实验室研究与探索, 2018, 37(4): 45-48. |
| [29] |
DANGELMAYR M A, REIMUS P W, WASSERMAN N L, et al. Laboratory column experiments and transport modeling to evaluate retardation of uranium in an aquifer downgradient of a uranium in situ recovery site[J]. Applied Geochemistry, 2017, 80: 1-13.
DOI URL |
| [30] | 张建明, 彭胜, 陈家军. 2,4-二氯苯酚在土壤中的吸附及其批实验与柱实验的分配系数比较[J]. 环境工程学报, 2012, 6(11): 4251-4256. |
| [31] |
MARAQA M. Effects of fundamental differences between batch and miscible displacement techniques on sorption distribution coefficient[J]. Environmental Geology, 2001, 41(1/2): 219-228.
DOI URL |
| [32] |
BARNETT M O, JARDINE P M, BROOKS S C, et al. Adsorption and transport of uranium(VI) in subsurface media[J]. Soil Science Society of America Journal, 2000, 64(3): 908-917.
DOI URL |
| [33] | Zheng C M, Bennett G D. Applied Contaminant Transport Modeling[J]. EOS Transactions American Geophysical Union, 2002, 77(48): 908-923. |
| [34] | 何宝南. 纳米乳化油制备及其在多孔介质中的迁移释放模拟实验[D]. 北京: 中国地质大学(北京), 2017. |
| [35] |
SCHWEICH D, SARDIN M, GAUDET J P. Measurement of a cation exchange isotherm from elution curves obtained in a soil column: preliminary results[J]. Soil Science Society of America Journal, 1983, 47(1): 32-37.
DOI URL |
| [36] | 张金利, 李宇. 碳纳米管-羟磷灰石对铅的吸附特性研究[J]. 环境科学, 2015, 36(7): 2554-2563. |
| [37] |
NOURI L, GHODBANE I, HAMDAOUI O, et al. Batch sorption dynamics and equilibrium for the removal of cadmium ions from aqueous phase using wheat bran[J]. Journal of Hazardous Materials. 2007, 149(1): 115-125.
DOI URL |
| [38] | GUELFO J L, WUNSCH A, MCCRAY J, et al. Subsurface transport potential of perfluoroalkyl acids (PFAAs): column experiments and modeling(Article)[J]. Journal of Contaminant Hydrology. 2020, 233: 103661. |
| [39] |
AVASARALA S, LICHTNER P C, ALI A M S, et al. Reactive transport of U and V from abandoned uranium mine wastes[J]. Environmental Science and Technology, 2017, 51(21): 12385-12393.
DOI URL |
| [40] | 孙占学, 马文洁, 刘亚洁, 等. 地浸采铀矿山地下水环境修复研究进展[J]. 地学前缘, 2021, 28(5): 215-225. |
| [41] | 昝金晶, 董一慧, 张卫民, 等. 铀在地下水系统中的赋存与迁移[J]. 有色金属(矿山部分), 2019, 71(6): 69-73, 77. |
| [1] | 鹿帅, 苏小四, 冯晓语, 孙超. 河水入渗过程中近岸带地下水中砷的形成与影响因素研究[J]. 地学前缘, 2022, 29(4): 455-467. |
| [2] | 郭巧娜, 赵岳, 周志芳, 林锦, 戴云峰, 李孟军. 人类活动影响下的龙口海岸带海底地下水排泄通量研究[J]. 地学前缘, 2022, 29(4): 468-479. |
| [3] | 王焰新, 李俊霞, 谢先军. 高碘地下水成因与分布规律研究[J]. 地学前缘, 2022, 29(3): 1-10. |
| [4] | 文冬光, 宋健, 刁玉杰, 张林友, 张福存, 张森琦, 叶成明, 朱庆俊, 史彦新, 金显鹏, 贾小丰, 李胜涛, 刘东林, 王新峰, 杨骊, 马鑫, 吴海东, 赵学亮, 郝文杰. 深部水文地质研究的机遇与挑战[J]. 地学前缘, 2022, 29(3): 11-24. |
| [5] | 邢世平, 郭华明, 吴萍, 胡学达, 赵振, 袁有靖. 化隆—循化盆地不同类型含水层组高氟地下水的分布及形成过程[J]. 地学前缘, 2022, 29(3): 115-128. |
| [6] | 侯国华, 高茂生, 叶思源, 赵广明. 黄河三角洲浅层地下水盐分来源及咸化过程研究[J]. 地学前缘, 2022, 29(3): 145-154. |
| [7] | 李海明, 李梦娣, 肖瀚, 刘学娜. 天津平原区浅层地下水水化学特征及碳酸盐风化碳汇研究[J]. 地学前缘, 2022, 29(3): 167-178. |
| [8] | 刘学娜, 李海明, 李梦娣, 章卫华, 肖瀚. 天津平原区加油站地下水石油烃污染特征及其生物降解机理研究[J]. 地学前缘, 2022, 29(3): 227-238. |
| [9] | 何宝南, 何江涛, 孙继朝, 王俊杰, 文冬光, 荆继红, 彭聪, 张昌延. 区域地下水污染综合评价研究现状与建议[J]. 地学前缘, 2022, 29(3): 51-63. |
| [10] | 郭华明, 高志鹏, 修伟. 地下水典型氧化还原敏感组分迁移转化的研究热点和趋势[J]. 地学前缘, 2022, 29(3): 64-75. |
| [11] | 廖福, 罗新, 谢月清, 易立新, 李海龙, 王广才. 氡(222Rn)在地下水-地表水相互作用中的应用研究进展[J]. 地学前缘, 2022, 29(3): 76-87. |
| [12] | 吕晓立, 郑跃军, 韩占涛, 李海军, 杨明楠, 张若琳, 刘丹丹. 城镇化进程中珠江三角洲地区浅层地下水中砷分布特征及成因[J]. 地学前缘, 2022, 29(3): 88-98. |
| [13] | 孙英, 周金龙, 杨方源, 纪媛媛, 曾妍妍. 塔里木盆地南缘绿洲带地下水砷氟碘分布及共富集成因[J]. 地学前缘, 2022, 29(3): 99-114. |
| [14] | 梁晓亮, 谭伟, 马灵涯, 朱建喜, 何宏平. 离子吸附型稀土矿床形成的矿物表/界面反应机制[J]. 地学前缘, 2022, 29(1): 29-41. |
| [15] | 黄冉笑, 王果胜, 袁国礼, 邱坤峰, Hounkpe Jechonias BIDOSSESSI. 伟晶质岩浆的同化混染与分离结晶(AFC)作用及铀成矿效应:以纳米比亚湖山铀矿为例[J]. 地学前缘, 2022, 29(1): 377-402. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||