

Earth Science Frontiers ›› 2022, Vol. 29 ›› Issue (3): 207-216.DOI: 10.13745/j.esf.2022.1.36
Previous Articles Next Articles
LIANG Kaixuan1,2( ), LIU Fei1,*(
), LIU Fei1,*( ), ZHANG Li3
), ZHANG Li3
Received:2021-12-06
Revised:2022-01-28
Online:2022-05-25
Published:2022-04-28
Contact:
LIU Fei
CLC Number:
LIANG Kaixuan, LIU Fei, ZHANG Li. Natural attenuation of perchlorate: A column experiment study[J]. Earth Science Frontiers, 2022, 29(3): 207-216.
| 水样 | 浓度/(mg·L-1) | pH | Eh | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cl- | K+ | Na+ | Ca2+ | Mg2+ | Fe(总) | |||||||
| 进水样 | 58.26 | 82.85 | 22.46 | 181.9 | 2.01 | 1.61 | 18.45 | 87.14 | 37.54 | 未检出 | 7.51 | 156 |
Table 1 Chemical component of groundwater- influent
| 水样 | 浓度/(mg·L-1) | pH | Eh | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cl- | K+ | Na+ | Ca2+ | Mg2+ | Fe(总) | |||||||
| 进水样 | 58.26 | 82.85 | 22.46 | 181.9 | 2.01 | 1.61 | 18.45 | 87.14 | 37.54 | 未检出 | 7.51 | 156 |
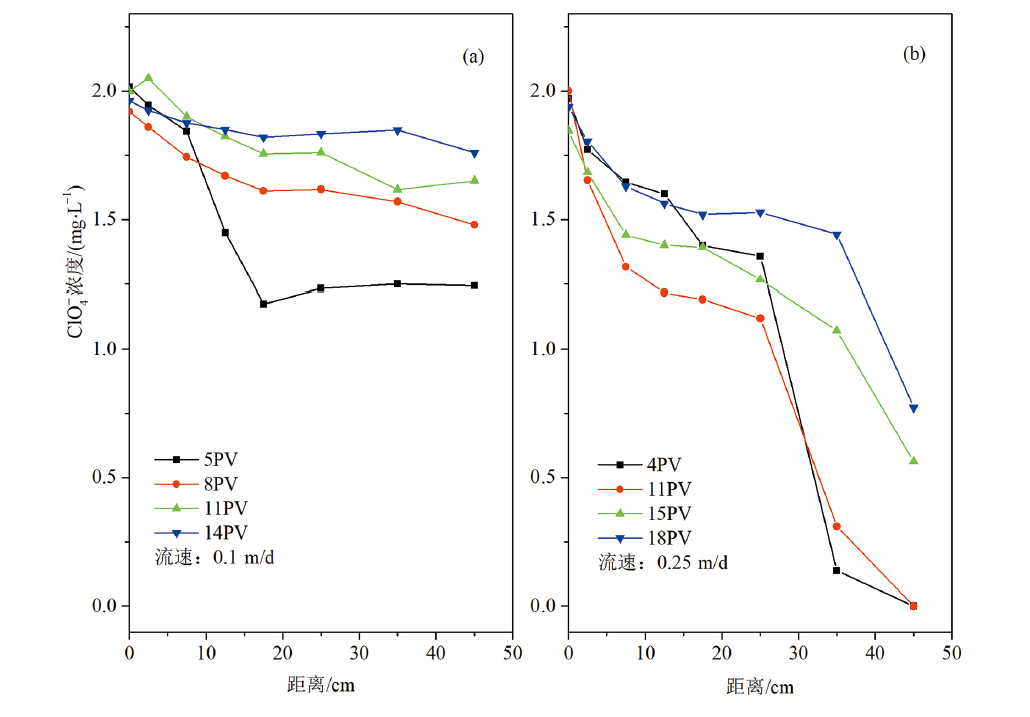
Fig.4 Change of perchlorate concentration with column distance under6 different pore volumes using effluents of (a) perchloate-deionized water and (b) perchlorate-groundwater solutions
| [1] |
SHIM Y H, HA S W, YOO E S, et al. Recovery of ammonium perchlorate from obsolete ammunition and its application in synthesis of lithium perchlorate[J]. Propellants Explosives Pyrotechnics, 2021, 46(4): 612-617.
DOI URL |
| [2] |
RAJAGOPALAN S, ANDERSON T, COX S, et al. Perchlorate in wet deposition across North America[J]. Environmental Science and Technology, 2009, 43(3): 616-222.
DOI URL |
| [3] | 郑雯静, 闻自强, 沈昊宇, 等. 高氯酸盐的来源、危害及其检测方法研究进展[J]. 环境科学与技术, 2018, 41(S1): 103-108. |
| [4] | 张丹, 段慧. 环境中高氯酸盐的污染、分析方法及去除技术的研究进展[J]. 西南农业学报, 2013, 26(6): 2648-2653. |
| [5] | 彭银仙. 高氯酸盐水环境行为特征研究[D]. 镇江: 江苏大学, 2011. |
| [6] |
LYNCH K L, JACKSON W A, REY K, et al. Evidence for biotic perchlorate reduction in naturally perchlorate-rich sediments of pilot valley basin, Utah[J]. Astrobiology, 2019, 19(5): 629-641.
DOI URL |
| [7] |
WAGNER H P, SUAREZ F X, PEPICH B V, et al. Challenges encountered in extending the sensitivity of US Environmental Protection Agency Method 314.0 for perchlorate in drinking water[J]. Journal of Chromatography A, 2004, 1039(1/2): 97-104.
DOI URL |
| [8] |
KRYNITSKY A J, NIEMANN R A, NORTRUP D A. Determination of perchlorate anion in foods by ion chromatography-tandem mass spectrometry[J]. Analytical Chemistry, 2004, 76(18): 5518-5522.
DOI URL |
| [9] |
SANCHEZ C A, KRIEGER R I, KHANDAKER N, et al. Accumulation and perchlorate exposure potential of lettuce produced in the Lower Colorado River region[J]. Journal of Agricultural and Food Chemistry, 2005, 53(13): 5479-5486.
DOI URL |
| [10] |
LUO W H, SASAKI K, HIRAJIMA T. Surfactant-modified montmorillonite by benzyloctadecyldimethylammonium chloride for removal of perchlorate[J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2015, 481:616-625.
DOI URL |
| [11] |
SHI Y L, ZHANG N, GAO J M, et al. Effect of fireworks display on perchlorate in air aerosols during the Spring Festival[J]. Atmospheric Environment, 2011, 45(6): 1323-1327.
DOI URL |
| [12] | 张萍, 史亚利, 蔡亚岐, 等. 离子色谱-质谱联用测定瓶装水中的高氯酸盐和溴酸盐[J]. 环境化学, 2007, 26(4): 544-546. |
| [13] | 史亚利, 高健民, 李鑫, 等. 浏阳河水、底泥和土壤中高氯酸盐的污染[J]. 环境化学, 2010, 29(3): 388-391. |
| [14] |
WU Q, ZHANG T, SUN H W, et al. Perchlorate in tap water, groundwater, surface waters, and bottled water from China and its association with other inorganic anions and with disinfection byproducts[J]. Archives of Environmental Contamination and Toxicology, 2010, 58(3): 543-550.
DOI URL |
| [15] |
TANG Y L, ZHONG B F, QU B, et al. Occurrence of perchlorate in groundwater, paired farmland soil, lettuce, and rhizosphere soil from Chengdu, China[J]. Environmental Science Processes and Impacts, 2017, 19(5): 752-757.
DOI URL |
| [16] |
KUMARATHILAKA P, OZE C, INDRARATNE S P, et al. Perchlorate as an emerging contaminant in soil, water and food[J]. Chemosphere, 2016, 150:667-677.
DOI URL |
| [17] |
RAO B, ANDERSON T A, REDDER A, et al. Perchlorate formation by ozone oxidation of aqueous chlorine/oxy-chlorine species: role of clxOy radicals[J]. Environmental Science and Technology, 2010, 44(8): 2961-2967.
DOI URL |
| [18] | 闫旭. 我国重点流域及重点地区饮用水中高氯酸盐污染水平调查研究[D]. 北京: 中国疾病预防控制中心, 2020. |
| [19] |
JACKSON W A, BÖHLKE J K, GU B H, et al. Isotopic composition and origin of indigenous natural perchlorate and co-occurring nitrate in the southwestern United States[J]. Environmental Science and Technology, 2010, 44(13): 4869-4876.
DOI URL |
| [20] |
HU J P, XIAN Y P, WU Y L, et al. Perchlorate occurrence in foodstuffs and water: analytical methods and techniques for removal from water - A review[J]. Food Chemistry, 2021, 360:130146.
DOI URL |
| [21] | 李冬桂, 农耀京, 吴凤, 等. 蔬菜水果中高氯酸盐检测及污染情况分析[J]. 食品科技, 2021, 46(2): 315-320. |
| [22] | 陈桂葵, 骆世明, 贺鸿志, 等. 高氯酸盐胁迫对水稻生长发育和养分吸收的影响[J]. 生态学报, 2014, 34(19): 5460-5469. |
| [23] | 方齐乐, 陈宝梁. 新型环境污染物高氯酸盐的环境化学行为、食品安全及健康风险[J]. 科学通报, 2013, 58(26): 2626-2642. |
| [24] |
CHEN Y N, ZHU Z, ZHAO Y, et al. Perchlorate in shellfish from South China Sea and implications for human exposure[J]. Marine Pollution Bulletin, 2021, 170:112672.
DOI URL |
| [25] | 田一媚, 宫智勇. 食品中高氯酸盐的污染现状及毒理作用研究进展[J]. 食品科学, 2020, 41(5): 276-281. |
| [26] |
GREER M A, GOODMAN G, PLEUS R C, et al. Health effects assessment for environmental perchlorate contamination: the dose response for inhibition of thyroidal radioiodine uptake in humans[J]. Environmental Health Perspectives, 2002, 110(9): 927-937.
DOI URL |
| [27] |
TIKKANEN M W. Development of a drinking water regulation for perchlorate in California[J]. Analytica Chimica Acta, 2006, 567(1): 20-25.
DOI URL |
| [28] | 于佳, 唐玄乐, 刘家仁. 高氯酸盐对人体健康影响的研究进展[J]. 环境与健康杂志, 2008, 25(7): 648-650. |
| [29] | 闻自强, 郑雯静, 沈昊宇, 等. 高氯酸盐的危害、水污染现状与去除技术研究进展[J]. 环境化学, 2019, 38(1): 209-216. |
| [30] |
MOTZER W E. Perchlorate: problems, detection, and solutions[J]. Environmental Forensics, 2001, 2(4): 301-311.
DOI URL |
| [31] |
SORIAL G A. The perchlorate dilemma in drinking water[J]. Journal of Environmental Engineering, 2004, 130(1): 1-2.
DOI URL |
| [32] | 蔡贤雷, 谢寅峰, 刘伟龙, 等. 高氯酸盐污染及修复的研究进展[J]. 生态学报, 2008, 28(11): 5592-5600. |
| [33] |
WANG C Y, DONG J, HU W, et al. Enhanced simultaneous removal of nitrate and perchlorate from groundwater by bioelectrochemical systems (BESs) with cathodic potential regulation[J]. Biochemical Engineering Journal, 2021, 173:108068.
DOI URL |
| [34] | 徐碧波. 生物碳及其改性材料对高氯酸盐去除性能及机理研究[D]. 长沙: 湖南大学, 2017. |
| [35] | 张红聘, 刘祥萱, 刘渊, 等. 高氯酸盐废水修复技术研究现状[J]. 化学推进剂与高分子材料, 2013, 11(3): 9-14. |
| [36] | 程丽杰, 高宁博, 楚华, 等. 高氯酸盐还原菌的代谢过程及应用研究进展[J]. 化工进展, 2020, 39(S2): 251-261. |
| [37] | LIANG S, SHI Q, GAO X Y, et al. Perchlorate removal by autotrophic bacteria associated with zero-valent iron: effect of calcium ions[J]. Journal of Chemical Technology and Biotechnology, 2015, 90(4): 722-729. |
| [38] |
ARTHUR R D, TORLAPATI J, SHIN K H, et al. Process control factors for continuous microbial perchlorate reduction in the presence of zero-valent iron[J]. Frontiers of Environmental Science and Engineering, 2014, 8(3): 386-393.
DOI URL |
| [39] | 高孟春, 赵从从, 张健, 等. 硫自养高氯酸盐还原菌培养与驯化[J]. 环境工程学报, 2014, 8(4): 1257-1261. |
| [40] | 邱华, 李佳璐, 龙雨涛, 等. 高氯酸盐高效降解菌的筛选及降解特性[J]. 水处理技术, 2015, 41(4): 58-61. |
| [41] | 吴春笃, 郭静, 许小红. 高氯酸盐降解菌的分离鉴定及特性研究[J]. 生态环境学报, 2010, 19(2): 281-285. |
| [42] | 时琼. 以零价铁为电子供体微生物协同去除地下水中高氯酸盐的研究[D]. 济南: 山东大学, 2012. |
| [43] | 王蕊, 刘菲, 陈鸿汉, 等. 电子供体对地下水中高氯酸盐生物去除的影响研究[J]. 环境科学学报, 2013, 33(11): 3060-3067. |
| [44] |
HAN W Y, FU F L, CHENG Z H, et al. Studies on the optimum conditions using acid-washed zero-valent iron/aluminum mixtures in permeable reactive barriers for the removal of different heavy metal ions from wastewater[J]. Journal of Hazardous Materials, 2016, 302:437-446.
DOI URL |
| [45] |
LONDON M R, WAHMAN D G, KATZ L E, et al. Zero-valent iron/biotic treatment system for perchlorate-contaminated water: lab-scale performance, modeling, and full-scale implications[J]. Journal of Environmental Engineering, 2013, 139(11): 1361-1367.
DOI URL |
| [46] |
SON A, LEE J, CHIU P C, et al. Microbial reduction of perchlorate with zero-valent iron[J]. Water Research, 2006, 40(10): 2027-2032.
DOI URL |
| [47] |
YU X Y, AMRHEIN C, DESHUSSES M A, et al. Perchlorate reduction by autotrophic bacteria in the presence of zero-valent iron[J]. Environmental Science and Technology, 2006, 40(4): 1328-1334.
DOI URL |
| [48] | 王小明. 几种亚稳态铁氧化物的结构、形成转化及其表面物理化学特性[D]. 武汉: 华中农业大学, 2015. |
| [49] | 张萌, 郑平. 铁氧化菌的类型、生境及富集培养方法[J]. 科技通报, 2012, 28(11): 72-76,152. |
| [50] | 卢玉琢. 铁锰氧化菌及硫酸盐还原菌在典型水环境中对金属腐蚀的影响研究[D]. 天津: 天津大学, 2011. |
| [51] | 王依依. 地下水高氯酸盐和硝酸盐复合污染的硫自养净化机制[D]. 郑州: 河南工业大学, 2018. |
| [52] |
LIU Y Y, PTACEK C J, BLOWES D W. Treatment of dissolved perchlorate, nitrate, and sulfate using zero-valent iron and organic carbon[J]. Journal of Environmental Quality, 2014, 43(3): 842-850.
DOI URL |
| [53] | 谢宇轩. 高氯酸盐及其与硝酸盐氮、氨氮混合污染的微生物降解研究[D]. 北京: 中国地质大学(北京), 2014. |
| [54] | 张波, 蒋霞, 李顺, 等. 零价铝对高氯酸盐污染物的去除[J]. 环境工程学报, 2016, 10(8): 4271-4276. |
| [55] | 谢宇轩, 刘菲, 关翔宇, 等. 基于功能基因表达的高氯酸盐与硝酸盐氮修复[J]. 环境工程学报, 2014, 8(4): 1423-1428. |
| [1] | HE Jiahui, MAO Hairu, XUE Yang, LIAO Fu, GAO Bai, RAO Zhi, YANG Yang, LIU Yuanyuan, WANG Guangcai. Variability in spatiotemporal groundwater nitrate concentrations in the northeast Ganfu Plain [J]. Earth Science Frontiers, 2024, 31(3): 360-370. |
| [2] | GUO Huaming, YIN Jiahong, YAN Song, LIU Chao. Distribution and source of nitrate in high-chromium groundwater in Jingbian, northern Shaanxi [J]. Earth Science Frontiers, 2024, 31(1): 384-399. |
| [3] | ZHANG Yuye, HE Jiangtao, DENG Lu, ZOU Hua, ZHANG Jingang, YANG Meiping. Effects of lomefloxacin and norfloxacin on the biological water denitrification process—an experimental study [J]. Earth Science Frontiers, 2022, 29(5): 497-507. |
| [4] | SHEN Xiaofang, WAN Yuyu, WANG Ligang, SU Xiaosi, DONG Weihong. Multiphase flow modeling of natural attenuation of volatile organic compounds (VOCs) in a petroleum contaminated sit [J]. Earth Science Frontiers, 2021, 28(5): 90-103. |
| [5] | MA Hong-Wen, SU Shuang-qing, LIU Gao, YANG Xue, BANG Hui, SHU Zi-Jian. Potassium resource and sustainable development of potash salt industry in China. [J]. Earth Science Frontiers, 2010, 17(1): 294-310. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||