

地学前缘 ›› 2023, Vol. 30 ›› Issue (4): 335-351.DOI: 10.13745/j.esf.sf.2023.2.42
杜佰松1,2,3( ), 朱光有2,*(
), 朱光有2,*( ), 刘舒飞1, 王业晗4, 于炳松3, 徐渴鑫3,5
), 刘舒飞1, 王业晗4, 于炳松3, 徐渴鑫3,5
收稿日期:2021-07-29
修回日期:2022-07-21
出版日期:2023-07-25
发布日期:2023-07-07
通讯作者:
*朱光有(1973—),男,博士,教授级高级工程师,博士生导师,主要从事油气地质与成藏地球化学等方面的研究工作。E⁃mail: zhuguangyou@petrochina.com.cn
作者简介:杜佰松(1992-),男,博士后,主要从事成因矿物学与找矿矿物学方面的研究工作。E-mail: BaisongDu@email.cugb.edu.cn
基金资助:
DU Baisong1,2,3( ), ZHU Guangyou2,*(
), ZHU Guangyou2,*( ), LIU Shufei1, WANG Yehan4, YU Bingsong3, XU Kexin3,5
), LIU Shufei1, WANG Yehan4, YU Bingsong3, XU Kexin3,5
Received:2021-07-29
Revised:2022-07-21
Online:2023-07-25
Published:2023-07-07
摘要:
碳酸盐岩约占显生宙沉积岩的20%,全球至少有60%已探明的石油储量赋存于碳酸盐岩储层中。方解石是普遍的碳酸盐矿物,其溶蚀对于地壳浅部次生孔隙的形成具有重要的意义。综述结果表明:方解石的生长主要受控于温度、压力、过饱和度、离子的种类及半径等因素;压力的降低、离子强度和过饱和度的增大有利于方解石的生长。方解石的溶蚀主要受温度、压力、p(CO2)分压、离子的种类和强度、盐度、pH值、介质流体组分、方解石表面Zeta电位及溶液相对于方解石的饱和程度等因素的影响;介质流体中的离子种类对方解石溶解所起的促进或抑制作用主要受控于方解石表面的Zeta电位。此外,本文还发现在方解石生长和溶蚀过程中,不同溶剂类型、离子的种类及浓度形成具有显著不同的微形貌,这使得通过对矿物微形貌的观察可以用来反演古介质流体的组分。本文以独特的视角对当前有关实验取得的不一致结果进行了深入剖析并提出了新的观点,着重对影响碳酸盐岩溶蚀的因素进行调研,寻找有利于碳酸盐岩溶蚀的最优解,进而为圈定优质储层提供可靠的理论依据。本文的另一个意义是便于后续相关研究能够悉知该领域的前沿热点及最新进展。
中图分类号:
杜佰松, 朱光有, 刘舒飞, 王业晗, 于炳松, 徐渴鑫. 浅析影响方解石生长和溶解的动力学因素及机制[J]. 地学前缘, 2023, 30(4): 335-351.
DU Baisong, ZHU Guangyou, LIU Shufei, WANG Yehan, YU Bingsong, XU Kexin. Key factors and mechanisms affecting calcite growth and dissolution-a critical review[J]. Earth Science Frontiers, 2023, 30(4): 335-351.
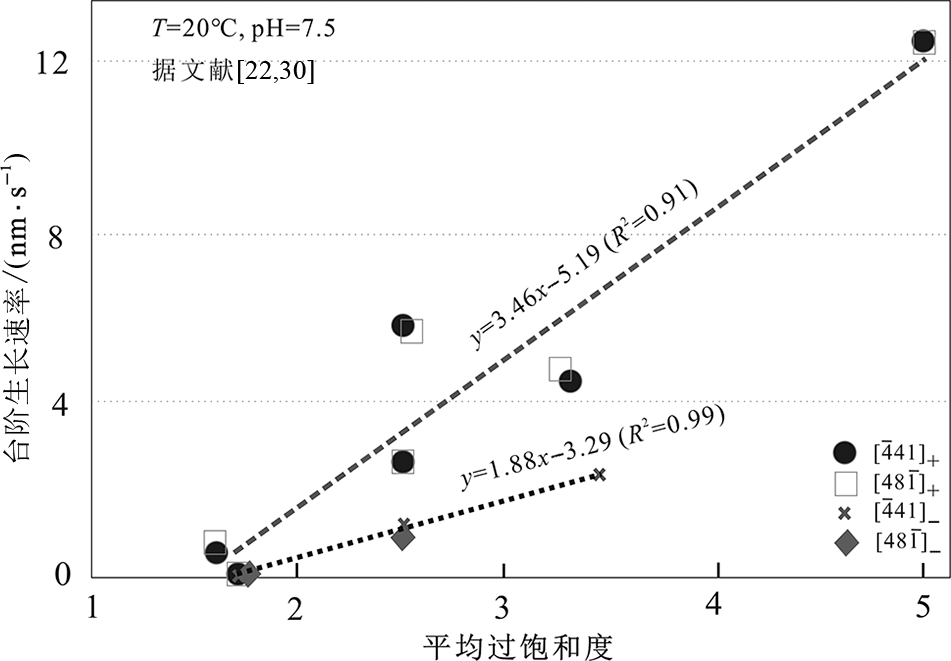
图1 方解石不同台阶的生长速率与过饱和度之间的关系 (据文献[22,30]整编)
Fig.1 Relationships between step velocity and calcite supersaturation at different cleavage faces. Data derived from [22,30].
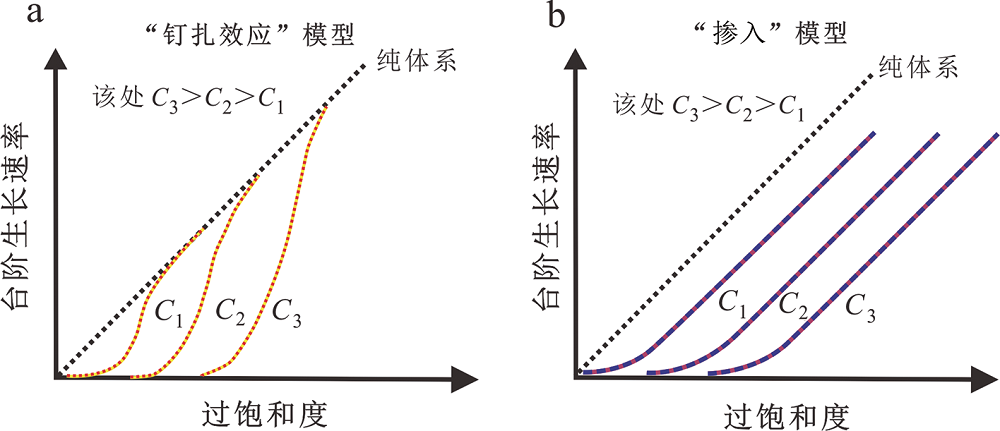
图2 台阶迁移速度与过饱和度、杂质元素浓度之间的关系 (底图据文献[26]修改) 图中C1,C2,C3代表杂质元素的浓度。
Fig.2 Dependency of step velocity on supersaturation according to different impurity models for calcite growth. C1,C2 and C3 are the impurity concentration in solution. Modified after [26].
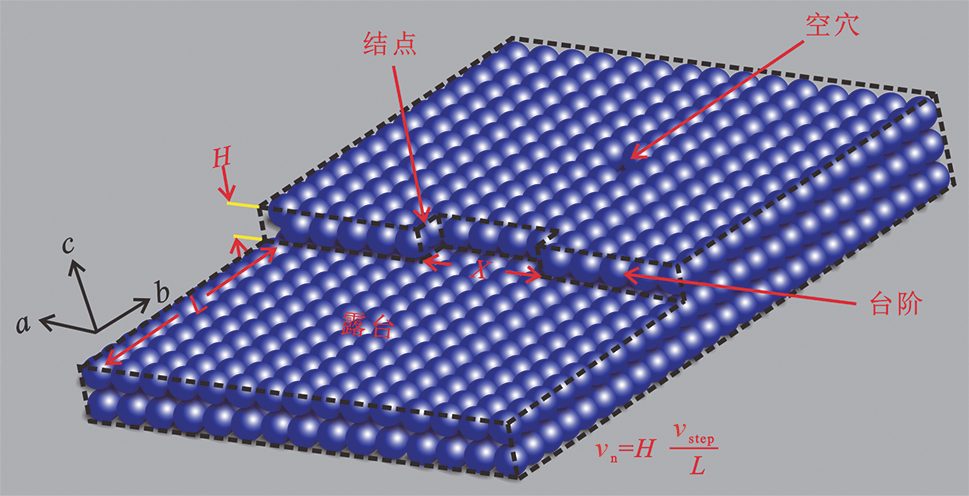
图3 晶体生长和溶解速率及其相关参数示意图 (据文献[33]修改) vn-晶体生长或溶蚀的整体速率;X-结点间距;H-台阶高度;vstep-台阶的迁移速度;L-露台宽度,也即台阶间距。
Fig.3 Sketch model of calcite step growth/dissolution relating calcite crystal growth/dissolution rates with bulk rate (vn). Modified after [33].
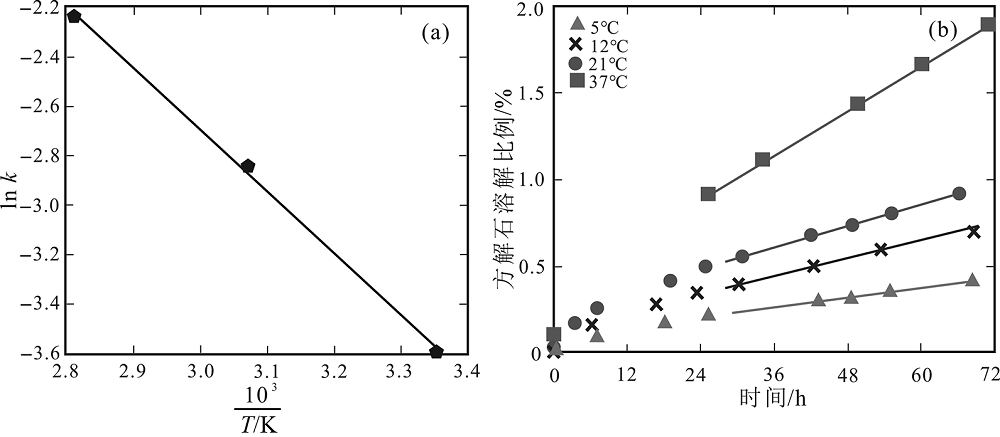
图4 温度对反应速率常数及方解石溶蚀的影响 (据文献[5,47]修改) a-ln k对1/T阿式投图,Ea值为20.9 kJ/mol;b-方解石的溶解与时间关系图。
Fig.4 Effect of temperature on calcite dissolution rate constant (a) and time course (b). Modified after [5,47].

图5 在CaCO3-CO2-H2O体系中温度(0~300 ℃)及p(CO2)分压对方解石溶解度的影响 (据文献[52,56-57]修改)
Fig.5 Influence of temperature and p(CO2) on calcite solubility in CaCO3-CO2-H2O system. Modified after [52,56-57].
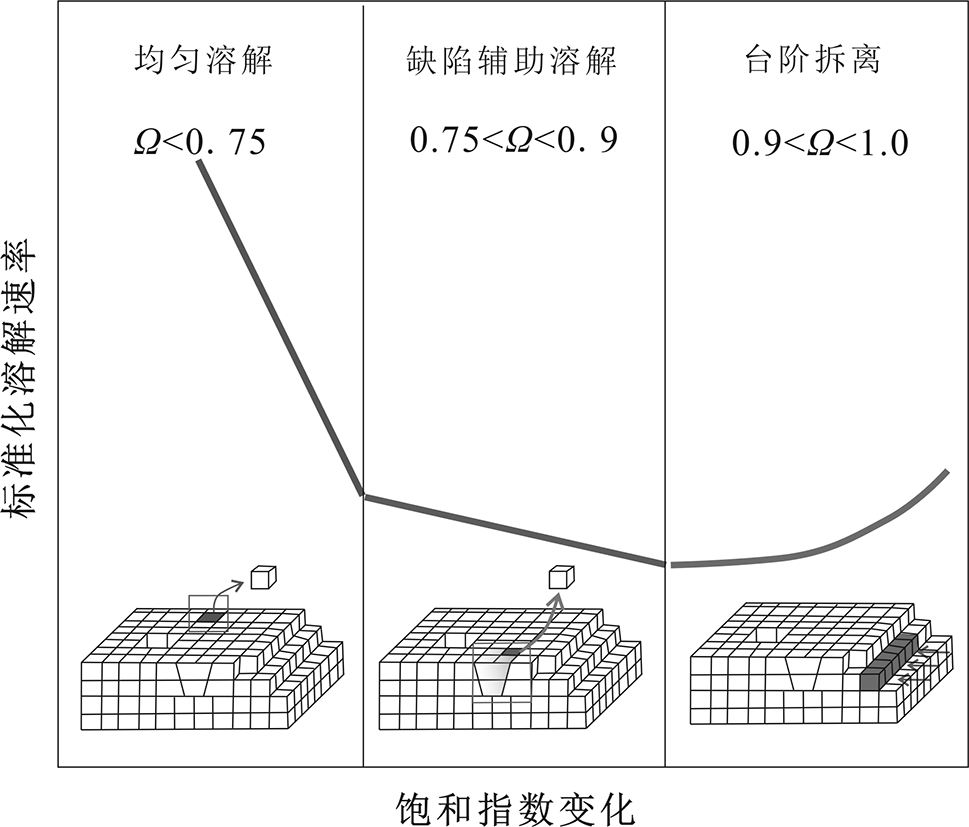
图7 方解石表面转变情况下不同溶解机制呈现的预期数据趋势 (据文献[47]修改)
Fig.7 Expected overall trend in calcite dissolution rate change under different dissolution mechanisms. Modified after [47].
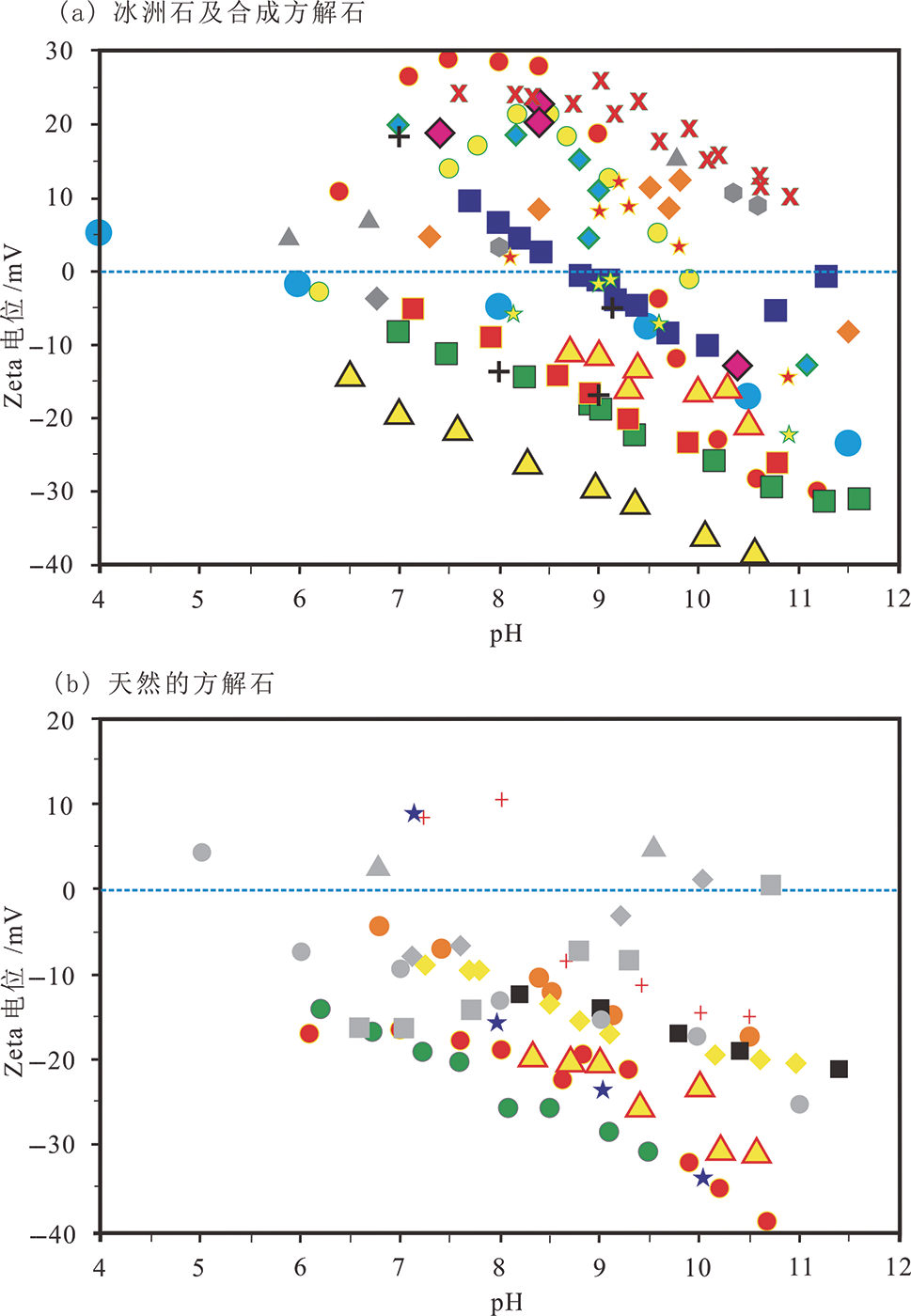
图8 不同类型的方解石表面电位随pH值变化趋势图 (据文献[88]修改)
Fig.8 Plots of Zeta potential of calcite vs. pH for different aqueous phosphate salts. (a) Iceland spar/synthetic calcite; (b) Natural calcite/carbonate rocks. Modified after [88].
| [1] |
DONG S J, BERELSON W M, ROLLINS N E, et al. Aragonite dissolution kinetics and calcite/aragonite ratios in sinking and suspended particles in the North Pacific[J]. Earth and Planetary Science Letters, 2019, 515: 1-12.
DOI URL |
| [2] |
TANG H M, XIAN H Y, HE H P, et al. Kinetics and mechanisms of the interaction between the calcite ($10\bar{1}4$) surface and Cu2+-bearing solutions[J]. Science of the Total Environment, 2019, 668: 602-616.
DOI URL |
| [3] |
POKROSKY O S, GOLUBEV S V, JORDAN G. Effect of organic and inorganic ligands on calcite and magnesite dissolution rates at 60 ℃ and 30 atm pCO2[J]. Chemical Geology, 2009, 265(1/2): 33-43.
DOI URL |
| [4] | 赵文智, 沈安江, 胡素云, 等. 中国碳酸盐岩储集层大型化发育的地质条件与分布特征[J]. 石油勘探与开发, 2012, 39(1): 1-12. |
| [5] |
GLEDHILL D K, MORSE J W. Calcite solubility in Na-Ca-Mg-Cl brines[J]. Chemical Geology, 2006, 233(3/4): 249-256.
DOI URL |
| [6] | JOSHI R M, SINGH K H. Petro-physics and rock physics of carbonate reservoirs[M]. New York: Springer, 2020: 4. |
| [7] | PANDYA N S, PANDYA J R. Etching of calcite[J]. Nature, 1959, 184(4690): 894-895. |
| [8] |
WATTS H. Etch pits on calcite cleavage faces[J]. Nature, 1959, 183(4657): 314.
DOI |
| [9] |
BERNER R A. Comparative dissolution characteristics of carbonate minerals in the presence and absence of aqueous magnesium ion[J]. American Journal of Science, 1967, 265(1): 45-70.
DOI URL |
| [10] |
MORSE J W. Dissolution kinetics of calcium carbonate in sea water: V.Effects of natural inhibitors and the position of the chemical lysocline[J]. American Journal of Science, 1974, 274(6): 638-647.
DOI URL |
| [11] | PLUMMER L N, PARKHURST D L, WIGLEY T M L. Critical review of the kinetics of calcite dissolution and precipitation[M]// ACS symposium series. Washington DC: American Chemical Society, 1979: 537-573. |
| [12] |
PLUMMER L N, WIGLEY T M L, PARKHURST D L. The kinetics of calcite dissolution in CO2-water systems at 5 degrees to 60 degrees C and 0.0 to 1.0 atm CO2[J]. American Journal of Science, 1978, 278(2): 179-216.
DOI URL |
| [13] |
SJÖBERG E L, RICKARD D T. The effect of added dissolved calcium on calcite dissolution kinetics in aqueous solutions at 25 ℃[J]. Chemical Geology, 1985, 49(4): 405-413.
DOI URL |
| [14] |
GUTJAHR A, DABRINGHAUS H, LACMANN R. Studies of the growth and dissolution kinetics of the CaCO3 polymorphs calcite and aragonite I. Growth and dissolution rates in water[J]. Journal of Crystal Growth, 1996, 158(3): 296-309.
DOI URL |
| [15] |
ALKATTAN M, OELKERS E H, DANDURAND J L, et al. An experimental study of calcite and limestone dissolution rates as a function of pH from -1 to 3 and temperature from 25 to 80 ℃[J]. Chemical Geology, 1998, 151(1/2/3/4): 199-214.
DOI URL |
| [16] |
CUBILLAS P, KÖHLER S, PRIETO M, et al. Experimental determination of the dissolution rates of calcite, aragonite, and bivalves[J]. Chemical Geology, 2005, 216(1/2): 59-77.
DOI URL |
| [17] |
GLEDHILL D K, MORSE J W. Dissolution kinetics of calcite in NaCl-CaCl2-MgCl2 brines at 25 ℃ and 1 bar pCO2[J]. Aquatic Geochemistry, 2004, 10(1/2): 171-190.
DOI URL |
| [18] |
RUIZ-AGUDO E, KOWACZ M, PUTNIS C V, et al. The role of background electrolytes on the kinetics and mechanism of calcite dissolution[J]. Geochimica et Cosmochimica Acta, 2010, 74(4): 1256-1267.
DOI URL |
| [19] |
RUIZ-AGUDO E, PUTNIS C V, JIMÉNEZ-LÓPEZ C, et al. An atomic force microscopy study of calcite dissolution in saline solutions: the role of magnesium ions[J]. Geochimica et Cosmochimica Acta, 2009, 73(11): 3201-3217.
DOI URL |
| [20] |
NEWTON R C, MANNING C E. Experimental determination of calcite solubility in H2O-NaCl solutions at deep crust/upper mantle pressures and temperatures: implications for metasomatic processes in shear zones[J]. American Mineralogist, 2002, 87(10): 1401-1409.
DOI URL |
| [21] | 陈勇, 王淼, 刘庆, 等. 盐效应和共同离子效应对方解石溶解度的影响及其地质意义[J]. 中国石油大学学报(自然科学版), 2016, 40(6): 33-39. |
| [22] |
GRATZ A J, HILLNER P E, HANSMA P K. Step dynamics and spiral growth on calcite[J]. Geochimica et Cosmochimica Acta, 1993, 57(2): 491-495.
DOI URL |
| [23] |
GRATZ A J, MANNE S, HANSMA P K. Atomic force microscopy of atomic-scale ledges and etch pits formed during dissolution of quartz[J]. Science, 1991, 251(4999): 1343-1346.
PMID |
| [24] |
ASTILLEROS J M, PINA C M, FERNÁNDEZ-DÍAZ L, et al. The effect of barium on calcite{$10\bar{1}4$} surfaces during growth[J]. Geochimica et Cosmochimica Acta, 2000, 64(17): 2965-2972.
DOI URL |
| [25] |
LEA A S, AMONETTE J E, BAER D R, et al. Microscopic effects of carbonate, manganese, and strontium ions on calcite dissolution[J]. Geochimica et Cosmochimica Acta, 2001, 65(3): 369-379.
DOI URL |
| [26] |
DAVIS K J, DOVE P M, DE YOREO J J. The role of Mg2+ as an impurity in calcite growth[J]. Science, 2000, 290(5494): 1134-1137.
DOI URL |
| [27] |
XU M, HIGGINS S R. Effects of magnesium ions on near-equilibrium calcite dissolution: step kinetics and morphology[J]. Geochimica et Cosmochimica Acta, 2011, 75(3): 719-733.
DOI URL |
| [28] |
WARD M D. Snapshots of crystal growth[J]. Science, 2005, 308(5728): 1566-1567.
DOI URL |
| [29] |
OHNESORGE F, BINNIG G. Trueatomic resolution by atomic force microscopy through repulsive and attractive forces[J]. Science, 1993, 260(5113): 1451-1456.
DOI URL |
| [30] |
HILLNER P E, GRATZ A J, MANNE S, et al. Atomic-scale imaging of calcite growth and dissolution in real time[J]. Geology, 1992, 20(4): 359-362.
DOI URL |
| [31] |
DOVE P M, HOCHELLA M F. Calcite precipitation mechanisms and inhibition by orthophosphate: in situ observations by Scanning Force Microscopy[J]. Geochimica et Cosmochimica Acta, 1993, 57(3): 705-714.
DOI URL |
| [32] |
TENG H H, DOVE P M, DE YOREO J J. Kinetics of calcite growth: surface processes and relationships to macroscopic rate laws[J]. Geochimica et Cosmochimica Acta, 2000, 64(13): 2255-2266.
DOI URL |
| [33] |
LARSEN K, BECHGAARD K, STIPP S L S. The effect of the Ca2+ to CO32- activity ratio on spiral growth at the calcite ($10\bar{1}4$) surface[J]. Geochimica et Cosmochimica Acta, 2010, 74(7): 2099-2109.
DOI URL |
| [34] |
REDDY M M, GAILLARD T D. Kinetics of calcium carbonate (calcite)-seeded crystallization: influence of solid/solution ratio on the reaction rate constant[J]. Journal of Colloid and Interface Science, 1981, 80(1): 171-178.
DOI URL |
| [35] |
WASYLENKI L E, DOVE P M, WILSON D S, et al. Nanoscale effects of strontium on calcite growth: an in situ AFM study in the absence of vital effects[J]. Geochimica et Cosmochimica Acta, 2005, 69(12): 3017-3027.
DOI URL |
| [36] |
NIELSEN M H, ALONI S, DE YOREOJ J. In situ TEM imaging of CaCO3 nucleation reveals coexistence of direct and indirect pathways[J]. Science, 2014, 345(6201): 1158-1162.
DOI URL |
| [37] |
HODKIN D J, STEWART D I, GRAHAM J T, et al. Enhanced crystallographic incorporation of strontium(II) ions into calcite via preferential adsorption at obtuse growth steps[J]. Crystal Growth and Design, 2018, 18(5): 2836-2843.
DOI URL |
| [38] |
KLASA J, RUIZ-AGUDO E, WANG L J, et al. An atomic force microscopy study of the dissolution of calcite in the presence of phosphate ions[J]. Geochimica et Cosmochimica Acta, 2013, 117: 115-128.
DOI URL |
| [39] |
PAQUETTE J, REEDER R J. Relationship between surface structure, growth mechanism, and trace element incorporation in calcite[J]. Geochimica et Cosmochimica Acta, 1995, 59(4): 735-749.
DOI URL |
| [40] |
RISTIC R I, DEYOREO J J, CHEW C M. Does impurity-induced step-bunching invalidate key assumptions of thecabrera-vermilyea model?[J]. Crystal Growth and Design, 2008, 8(4): 1119-1122.
DOI URL |
| [41] |
VAN ENCKEVORT W J P, VAN DEN BERG A C J F. Impurity blocking of crystal growth: a Monte Carlo study[J]. Journal of Crystal Growth, 1998, 183(3): 441-455.
DOI URL |
| [42] |
ARVIDSON R S, COLLIER M, DAVIS K J, et al. Magnesium inhibition of calcite dissolution kinetics[J]. Geochimicaet Cosmochimica Acta, 2006, 70(3): 583-594.
DOI URL |
| [43] |
TENG H H, DOVE P M, ORME C A, et al. Thermodynamics of calcite growth:baseline for understanding biomineral formation[J]. Science, 1998, 282(5389): 724-727.
DOI URL |
| [44] |
ZHANG Y, DAWE R. The kinetics of calcite precipitation from a high salinity water[J]. Applied Geochemistry, 1998, 13(2): 177-184.
DOI URL |
| [45] |
BISCHOFF J L. Kinetics of calcite nucleation:magnesium ion inhibition and ionic strength catalysis[J]. Journal of Geophysical Research, 1968, 73(10): 3315-3322.
DOI URL |
| [46] |
TENG H H. Controls by saturation state on etch pit formation during calcite dissolution[J]. Geochimica et Cosmochimica Acta, 2004, 68(2):253-262.
DOI URL |
| [47] |
NAVIAUX J D, SUBHAS A V, ROLLINS N E, et al. Temperature dependence of calcite dissolution kinetics in seawater[J]. Geochimica et Cosmochimica Acta, 2019, 246: 363-384.
DOI |
| [48] |
JEAN C. The alkaline dissolution rate of calcite[J]. Journal of Physical Chemistry Letters, 2016, 7(13): 2376-2380.
DOI PMID |
| [49] |
VINSON M, LÜTTGE A. Multiple length-scale kinetics: an integrated study of calcite dissolution rates and strontium inhibition[J]. American Journal of Science, 2005, 305(2): 119-146.
DOI URL |
| [50] |
SJÖBERG E L, RICKARD D T. Temperature dependence of calcite dissolution kinetics between 1 and 62 ℃ at pH 2.7 to 8.4 in aqueous solutions[J]. Geochimica et Cosmochimica Acta, 1984, 48(3): 485-493.
DOI URL |
| [51] |
FINNERAN D W, MORSE J W. Calcite dissolution kinetics in saline waters[J]. Chemical Geology, 2009, 268(1): 137-146.
DOI URL |
| [52] |
ELLIS A J. The solubility of calcite in carbon dioxide solutions[J]. American Journal of Science, 1959, 257(5): 354-365.
DOI URL |
| [53] | SJÖBERG E L. Kinetics and mechanism of calcite dissolution in aqueous solutions at low temperatures[J]. Stockholm Contributions in Geology, 1978, 32: 1-92. |
| [54] |
MACINNISIAN N, BRANTLEY S L. The role of dislocations and surface morphology in calcite dissolution[J]. Geochimica et Cosmochimica Acta, 1992, 56(3): 1113-1126.
DOI URL |
| [55] |
FREDD C N, FOGLER H S. The kinetics of calcite dissolution in acetic acid solutions[J]. Chemical Engineering Science, 1998, 53(22): 3863-3874.
DOI URL |
| [56] |
MILLER J P. A portion of the system calcium carbonate-carbon dioxide-water, with geological implications[J]. American Journal of Science, 1952, 250(3): 161-203.
DOI URL |
| [57] |
SEGNIT E R, HOLLAND H D, BISCARDI C J. The solubility of calcite in aqueous solutions: I.The solubility of calcite in water between 75 ℃ and 200 ℃ at CO2 pressures up to 60 atm[J]. Geochimica et Cosmochimica Acta, 1962, 26(12): 1301-1331.
DOI URL |
| [58] |
SHARP W E, KENNEDY G C. Thesystem CaO-CO2-H2O in the two-phase region calcite + aqueous solution[J]. The Journal of Geology, 1965, 73(2): 391-403.
DOI URL |
| [59] |
CACIAGLI N C, MANNING C E. The solubility of calcite in water at 6-16 kbar and 500-800 ℃[J]. Contributions to Mineralogy and Petrology, 2003, 146: 275-285.
DOI URL |
| [60] | WALTHER J V, LONG M I. Experimental determination of calcite solubilities in supercritical H2O[C]// Fifth international symposium on water-rock interaction. Reykjavik, Iceland: National Energy Authority, 1986, 5: 609-611. |
| [61] |
FEIN J B, WALTHER J V. Calcite solubility and speciation in supercritical NaCl-HCl aqueous fluids[J]. Contributions to Mineralogy and Petrology, 1989, 103(3): 317-324.
DOI URL |
| [62] | 范明, 胡凯, 蒋小琼, 等. 酸性流体对碳酸盐岩储层的改造作用[J]. 地球化学, 2009, 38(1): 20-26. |
| [63] |
SALEM M R, MANGOOD A H, HAMDONA S K. Dissolution of calcite crystals in the presence of some metal ions[J]. Journal of Materials Science, 1994, 29(24): 6463-6467.
DOI URL |
| [64] |
KEIR R S. The dissolution kinetics of biogenic calcium carbonates in seawater[J]. Geochimica et Cosmochimica Acta, 1980, 44(2): 241-252.
DOI URL |
| [65] |
LASAGA A C, LUTTGE A. Variation of crystal dissolution rate based on a dissolution stepwave model[J]. Science, 2001, 291(5512): 2400-2404.
PMID |
| [66] |
KURGANSKAYA I, LUTTGE A. Kinetic Monte Carlo approach to study carbonate dissolution[J]. The Journal of Physical Chemistry C, 2016, 120(12): 6482-6492.
DOI URL |
| [67] |
BOUDREAU B P. Carbonate dissolution rates at the deep ocean floor[J]. Geophysical Research Letters, 2013, 40(4): 744-748.
DOI URL |
| [68] |
PETERSON M N A. Calcite: rates of dissolution in a vertical profile in the central Pacific[J]. Science, 1966, 154(3756): 1542-1544.
PMID |
| [69] |
HONJO S, EREZ J. Dissolution rates of calcium carbonate in the deep ocean: an in situ experiment in the North Atlantic Ocean[J]. Earth and Planetary Science Letters, 1978, 40: 287-300.
DOI URL |
| [70] |
FUKUHARA T, TANAKA Y, IOKA N, et al. An in situ experiment of calcium carbonate dissolution in the central Pacific Ocean[J]. International Journal of Greenhouse Gas Control, 2008, 2(1): 78-88.
DOI URL |
| [71] |
SUBHAS A V, ADKINS J F, ROLLINS N E, et al. Catalysis and chemical mechanisms of calcite dissolution in seawater[J]. Proceedings of the National Academy of Sciences of the United States of America, 2017, 114(31): 8175-8180.
DOI PMID |
| [72] |
DONG S, SUBHAS A V, ROLLINS N E, et al. A kinetic pressure effect on calcite dissolution in seawater[J]. Geochimica et Cosmochimica Acta, 2018, 238: 411-423.
DOI URL |
| [73] |
BLUM A E, YUND R A, LASAGA A C. The effect of dislocation density on the dissolution rate of quartz[J]. Geochimica et Cosmochimica Acta, 1990, 54(2): 283-297.
DOI URL |
| [74] |
WALTER L M, BURTON E A. The effect of orthophosphate on carbonate mineral dissolution rates in seawater[J]. Chemical Geology, 1986, 56(3/4): 313-323.
DOI URL |
| [75] |
BERNER R A, MORSE J W. Dissolution kinetics of calcium carbonate in sea water: IV.Theory of calcite dissolution[J]. American Journal of Science, 1974, 274(2): 108-134.
DOI URL |
| [76] |
NAVIAUX J D, SUBHAS A V, DONG S J, et al. Calcite dissolution rates in seawater:lab vs. in situ measurements and inhibition by organic matter[J]. Marine Chemistry, 2019, 215: 103684.
DOI URL |
| [77] |
GUTJAHR A, DABRINGHAUS H, LACMANN R. Studies of the growth and dissolution kinetics of the CaCO3 polymorphs calcite and aragonite: II.The influence of divalent cation additives on the growth and dissolution rates[J]. Journal of Crystal Growth, 1996, 158(3): 296-309.
DOI URL |
| [78] |
BUHMANN D, DREYBRODT W. Calcite dissolution kinetics in the system H2O-CO2-CaCO3 with participation of foreign ions[J]. Chemical Geology, 1987, 64(1/2): 89-102.
DOI URL |
| [79] |
COMPTON R G, BROWN C A. The inhibition of calcite dissolution/precipitation: Mg2+ cations[J]. Journal of Colloid and Interface Science, 1994, 165(2): 445-449.
DOI URL |
| [80] |
ALKATTAN M, OELKERS E H, DANDURAND J L, et al. An experimental study of calcite dissolution rates at acidic conditions and 25 ℃ in the presence of NaPO3 and MgCl2[J]. Chemical Geology, 2002, 190(1/2/3/4): 291-302.
DOI URL |
| [81] | SABBIDES T G, KOUTSOUKOS P G. The dissolution of calcium carbonate in the presence of magnesium and inorganic orthophosphate[M]// Mineral scale formation and inhibition. Boston: Springer, 1995: 73-86. |
| [82] | PARKS G A. Adsorption in the marine environment[M]// RILEY P, SKIRROW G. Chemical oceanography. San Francisco: Academic Press, 1975: 241-308. |
| [83] |
DE KANEL J, MORSE J W. The chemistry of orthophosphate uptake from seawater on to calcite and aragonite[J]. Geochimica et Cosmochimica Acta, 1978, 42(9): 1335-1340.
DOI URL |
| [84] |
MORSE J W, MACKENZIE F T. Geochemical constraints on CaCO3 transport in subsurface sedimentary environments[J]. Chemical Geology, 1993, 105(1/2/3): 181-196.
DOI URL |
| [85] |
MILLERO F, HUANG F, ZHU X R, et al. Adsorption and desorption of phosphate on calcite and aragonite in seawater[J]. Aquatic Geochemistry, 2001, 7(1): 33-56.
DOI URL |
| [86] |
THOMPSON D W, POWNALL P G. Surface electrical-properties of calcite[J]. Journal of Colloid and Interface Science, 1989, 131(1): 74-82.
DOI URL |
| [87] |
KASHA A, AL-HASHIM H, ABDALLAH W, et al. Effect of Ca2+, Mg2+ and SO42- ions on the Zeta potential of calcite and dolomite particles aged with stearic acid[J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2015, 482: 290-299.
DOI URL |
| [88] |
AL MAHROUQI D, VINOGRADOV J, JACKSON M D. Zeta potential of artificial and natural calcite in aqueous solution[J]. Advances in Colloid and Interface Science, 2017, 240: 60-76.
DOI PMID |
| [89] |
SIFFERT D, FIMBEL P. Parameters affecting the sign and magnitude of the eletrokinetic potential of calcite[J]. Colloids and Surfaces, 1984, 11(3/4): 377-389.
DOI URL |
| [90] |
POKROVSKY O, GOLUBEV S, SCHOTT J. Dissolution kinetics of calcite, dolomite and magnesite at 25 ℃ and 0 to 50 atm pCO2[J]. Chemical Geology, 2005(3/4), 217: 239-255.
DOI URL |
| [91] |
ZUDDAS P, MUCCI A. Kinetics ofcalcite precipitation from seawater: II.The influence of the ionic strength[J]. Geochimica et Cosmochimica Acta, 1998, 62(5): 757-766.
DOI URL |
| [92] | LIPPMANN F. Sedimentary carbonate minerals[J]. Chemical Geology, 1973, 12: 407-407. |
| [93] |
ARVIDSON R S, MACKENZIE F T. The dolomite problem: control of precipitation kinetics by temperature and saturation state[J]. American Journal of Science, 1999, 299: 257-288.
DOI URL |
| [94] |
LIANG Y, BAER D R, MCCOY J M, et al. Dissolution kinetics at the calcite-water interface[J]. Geochimica et Cosmochimica Acta, 1996, 60(23): 4883-4887.
DOI URL |
| [95] |
XU M, HU X M, KNAUSS K G, et al. Dissolution kinetics of calcite at 50 to 70 ℃: an atomic force microscopic study under near-equilibrium conditions[J]. Geochimica et Cosmochimica Acta, 2010, 74: 4285-4297.
DOI URL |
| [96] |
TENG H H, DOVE P M, DE YOREO J J. Reversed calcite morphologies induced by microscopic growth kinetics: insight into biomineralization[J]. Geochimica et Cosmochimica Acta, 1999, 63(17): 2507-2512.
DOI URL |
| [97] |
杨海军, 陈永权, 田军, 等. 塔里木盆地轮探1井超深层油气勘探重大发现与意义[J]. 中国石油勘探, 2020, 25(2): 62-72.
DOI |
| [1] | 樊太亮, 高志前, 吴俊. 塔里木盆地深层碳酸盐岩建造-改造作用与多类型储层有序性分布[J]. 地学前缘, 2023, 30(4): 1-18. |
| [2] | 徐大良, 邓新, 彭练红, 田洋, 金巍, 金鑫镖. 大别山碰撞造山带俯冲盘陆壳基底组成:白垩纪脉岩捕获/继承锆石的证据[J]. 地学前缘, 2023, 30(4): 299-316. |
| [3] | 杨德彬, 鲁新便, 高志前, 曹飞, 汪彦, 鲍典, 李生青. 塔北深层海相碳酸盐岩断溶体成藏认识及油藏特征[J]. 地学前缘, 2023, 30(4): 43-50. |
| [4] | 吴晨, 陈宣华, 丁林. 祁连造山带构造演化与新生代变形历史[J]. 地学前缘, 2023, 30(3): 262-281. |
| [5] | 刘震, 朱茂林, 潘高峰, 夏鲁, 卢朝进, 刘明洁, 刘静静, 侯英杰. 鄂尔多斯盆地中南部地区延长组砂岩溶蚀增孔模型的建立与应用[J]. 地学前缘, 2023, 30(2): 96-108. |
| [6] | 包洪平, 王前平, 闫伟, 蔡郑红, 郑杰, 魏柳斌, 黄正良, 郭玮. 鄂尔多斯盆地中东部奥陶系碳酸盐岩-膏盐岩体系沉积特征与天然气成藏潜力[J]. 地学前缘, 2023, 30(1): 30-44. |
| [7] | 王根久, 宋新民, 刘波, 石开波, 刘航宇. 伊拉克A油田白垩系孔隙型生屑灰岩高渗透层成因及分布特征[J]. 地学前缘, 2022, 29(5): 483-496. |
| [8] | 徐盛林, 丁伟翠, 陈宣华, 李廷栋, 韩乐乐, 刘勇, 马飞宙, 王叶. 西准噶尔晚古生代地壳组成与生长:来自Sr-Nd-Pb同位素填图的证据[J]. 地学前缘, 2022, 29(2): 261-280. |
| [9] | 张卫民, 王振, 钱程, 郭亚丹, 刘海燕. 方解石负载羟基磷灰石复合材料去除水中铀离子的PRB活性介质研究[J]. 地学前缘, 2021, 28(5): 175-185. |
| [10] | 魏巍, 朱筱敏, 朱世发, 何明薇, 孙书洋, 王名巍. 二连盆地额仁淖尔凹陷下白垩统湖相云质岩优质储层特征及控制因素[J]. 地学前缘, 2021, 28(1): 214-224. |
| [11] | 刘伟, 黄擎宇, 白莹, 石书缘. 同生-准同生期大气淡水溶蚀对微生物碳酸盐岩储层的控制作用:以塔里木盆地下寒武统为例[J]. 地学前缘, 2021, 28(1): 225-234. |
| [12] | 李亭昕, 蔡永丰, 刘彦广, 刘桂宏, 张宏亮, 秦祥熙. 献县地热田碳酸盐岩热储示踪试验与模拟[J]. 地学前缘, 2020, 27(1): 152-158. |
| [13] | 张忠义. 大巴山西北缘浅层次叠加褶皱变形分析:以镇巴县简池地区为例[J]. 地学前缘, 2019, 26(2): 1-15. |
| [14] | 何治亮,云露,尤东华,彭守涛,张宏,王康宁,钱一雄,焦存礼,张继标. 塔里木盆地阿满过渡带超深层碳酸盐岩储层成因与分布预测[J]. 地学前缘, 2019, 26(1): 13-21. |
| [15] | 王涛,侯增谦. 同位素填图与深部物质探测(Ⅰ):揭示岩石圈组成演变与地壳生长[J]. 地学前缘, 2018, 25(6): 1-19. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||