

地学前缘 ›› 2024, Vol. 31 ›› Issue (2): 20-30.DOI: 10.13745/j.esf.sf.2023.9.8
收稿日期:2023-08-18
修回日期:2023-09-01
出版日期:2024-03-25
发布日期:2024-04-18
通讯作者:
*郭朝晖(1971—),男,博士,教授,主要研究方向为土壤重金属污染控制与修复。E-mail: 作者简介:梁慧芝(1999—),女,硕士研究生,主要研究方向为土壤重金属污染控制与修复。E-mail: 213512174@csu.edu.cn
基金资助:
LIANG Huizhi( ), GUO Zhaohui*(
), GUO Zhaohui*( ), ZHANG Yunxia, XU Rui
), ZHANG Yunxia, XU Rui
Received:2023-08-18
Revised:2023-09-01
Online:2024-03-25
Published:2024-04-18
摘要:
含砷尾矿中砷(As)及伴生元素铊(Tl)等毒性元素易向周边水和土壤介质中迁移,揭示尾矿中毒性元素的矿相特征及其释放机制具有重要意义。本研究以某雄黄矿区含砷尾矿为研究对象,结合化学分析、矿物学表征等手段,通过静态浸出实验探究含砷尾矿中As和Tl的矿相特征及其释放机制。研究结果表明,含砷尾矿中As和Tl的环境行为明显受到其矿相特征及赋存形态影响。矿物学分析结果显示尾矿中As的主要矿物相为砷铂矿和砷铁矿,矿物解离度表明砷矿物处于风化状态,As释放风险较高;而Tl则以伴生元素形式存在于Ca、Mn和Mg等矿物相中,这些矿物的沉淀溶解控制着尾矿中Tl的释放。尾矿中As主要以铁锰氧化物结合态和有机结合态存在,Tl主要以铁锰氧化物结合态和残渣态存在。酸性浸出条件下金属的活性态比例增高,可交换态As由0.29%上升至1.67%,可交换态Tl从5.46%升高至8.67%;尾矿中As释放加强而Tl受到抑制,表明尾矿中As与Tl释放存在竞争关系。As的浸出符合双常数模型,为多因素控制的物理和化学过程;Tl的浸出符合抛物线扩散模型,由结构掺入和表面吸附等扩散机制控制。本研究明确了含砷尾矿As和Tl的释放特性、化学形态转变及矿物相特征,为含砷尾矿中As和Tl污染风险控制提供了科学依据。
中图分类号:
梁慧芝, 郭朝晖, 张云霞, 徐锐. 含砷尾矿中砷铊矿相特征及其释放机制[J]. 地学前缘, 2024, 31(2): 20-30.
LIANG Huizhi, GUO Zhaohui, ZHANG Yunxia, XU Rui. Mineralogical characteristics and release mechanism of arsenic-thallium from As-bearing tailings[J]. Earth Science Frontiers, 2024, 31(2): 20-30.
| 元素 | wB/% | 元素 | wB/% |
|---|---|---|---|
| As | 0.16 | Mn | 0.018 |
| Tl | 0.000 566 | Pb | 0.005 |
| Ca | 22.3 | Sr | 0.012 |
| Mg | 11.2 | Al | 0.26 |
| Fe | 0.21 | S | 0.038 |
表1 含砷尾矿主要元素组成和重金属含量
Table 1 Major element composition and heavy metal content in As-bearing tailings
| 元素 | wB/% | 元素 | wB/% |
|---|---|---|---|
| As | 0.16 | Mn | 0.018 |
| Tl | 0.000 566 | Pb | 0.005 |
| Ca | 22.3 | Sr | 0.012 |
| Mg | 11.2 | Al | 0.26 |
| Fe | 0.21 | S | 0.038 |
| 矿物种类 | 化学式 | wB/% | 矿物种类 | 化学式 | wB/% | ||
|---|---|---|---|---|---|---|---|
| Dolomite | 白云石 | CaMg(CO3)2 | 82.12 | Angelellite | 砷铁矿 | Fe4(AsO4)2O3 | 0.41 |
| Calcite | 方解石 | CaCO3 | 4.87 | Kaolinite | 高岭石 | Al2Si2O5(OH)4 | 0.18 |
| Gedrite | 铝直闪石 | Mg5A12(Si6A12O22)(OH)2 | 0.23 | Albite | 钠长石 | Na2OAl2O36SiO2 | 0.17 |
| Sperrylite | 砷铂矿 | PtAs2 | 0.23 | Ferritschermakite | 铁镁钙闪石 | Ca2Fe2MgA13Si7O22(OH)2 | 0.8 |
| Orthoclase | 正长石 | KAlSi3O8 | 1.25 | Quartz | 石英 | SiO2 | 2.88 |
| Iron | 铁 | Fe | 0.09 | ||||
表2 含砷尾矿的主要矿物相组成
Table 2 Major minerals in As-bearing tailings
| 矿物种类 | 化学式 | wB/% | 矿物种类 | 化学式 | wB/% | ||
|---|---|---|---|---|---|---|---|
| Dolomite | 白云石 | CaMg(CO3)2 | 82.12 | Angelellite | 砷铁矿 | Fe4(AsO4)2O3 | 0.41 |
| Calcite | 方解石 | CaCO3 | 4.87 | Kaolinite | 高岭石 | Al2Si2O5(OH)4 | 0.18 |
| Gedrite | 铝直闪石 | Mg5A12(Si6A12O22)(OH)2 | 0.23 | Albite | 钠长石 | Na2OAl2O36SiO2 | 0.17 |
| Sperrylite | 砷铂矿 | PtAs2 | 0.23 | Ferritschermakite | 铁镁钙闪石 | Ca2Fe2MgA13Si7O22(OH)2 | 0.8 |
| Orthoclase | 正长石 | KAlSi3O8 | 1.25 | Quartz | 石英 | SiO2 | 2.88 |
| Iron | 铁 | Fe | 0.09 | ||||

图1 尾矿中As、Ca、Fe和Mg的矿物赋存形态(a)及其矿物解离度(b)
Fig.1 Occurrence of elements As, Ca, Fe, Mg in major minerals (a) and dissolution profiles of major minerals in As-bearing tailings
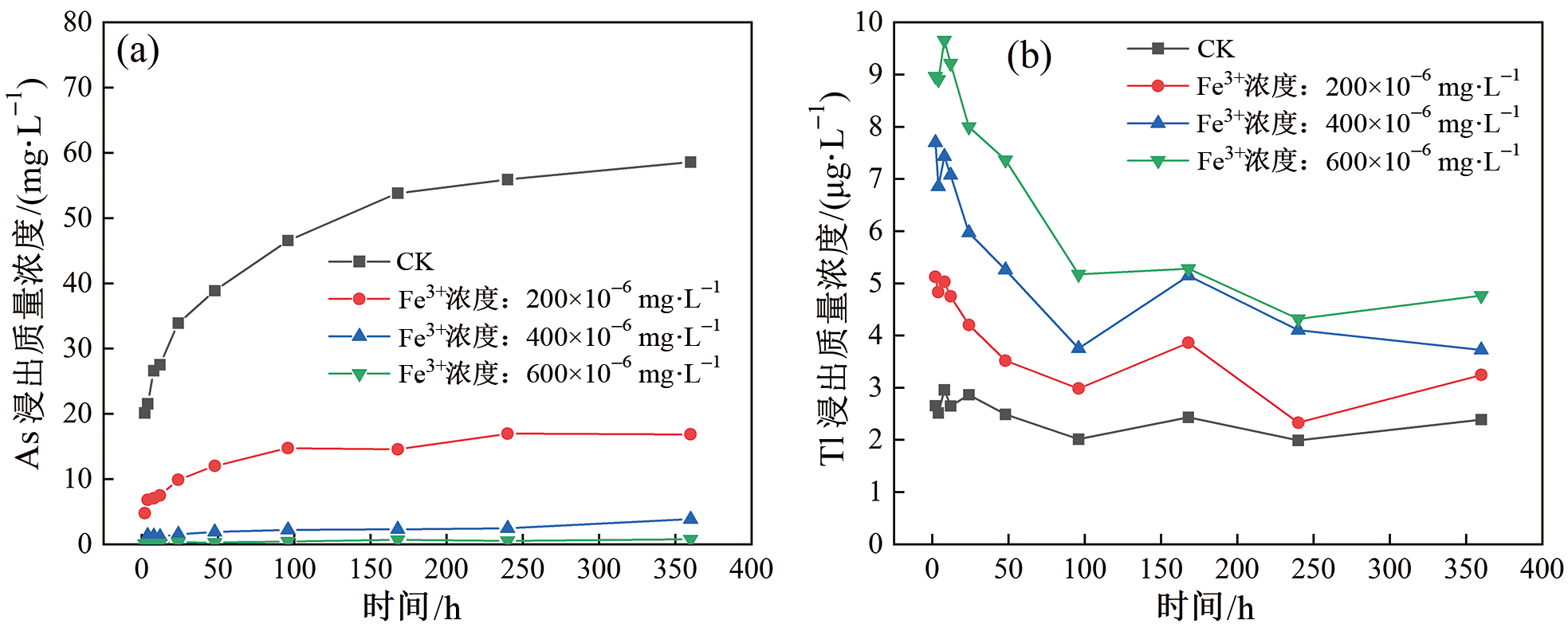
图3 不同Fe3+浓度下浸出液中As(a)和Tl(b)的浓度变化图
Fig.3 Temporal profiles of As (a) and Tl (b) concentrations in tailing leachates under different Fe3+ concentrations

图5 不同处理组浸出液的Ca(a, b, c)和Mg(d, e, f)浓度变化(a和d: pH; b和e: Fe3+浓度; c和f: 粒径)
Fig.5 Temporal profiles of Ca (a, b, c) and Mg (d, e, f) concentrations in tailing leachates under different conditions
| 元素 | 浸出环境 | 符合模型 | 参数 | ||
|---|---|---|---|---|---|
| a | b | R2 | |||
| As | pH=3.0 | 双常数方程 | 2.754 1 | 0.242 6 | 0.997 0 |
| pH=4.5 | 双常数方程 | 2.345 1 | 0.311 3 | 0.990 2 | |
| pH=7.0 | 双常数方程 | 2.418 9 | 0.297 4 | 0.993 4 | |
| pH=9.0 | 双常数方程 | 2.445 6 | 0.294 8 | 0.993 2 | |
| Fe3+浓度:200×10-6 mg·L-1 | Elovich方程 | 2.513 2 | 2.474 5 | 0.971 9 | |
| Fe3+浓度:400×10-6 mg·L-1 | 抛物线扩散方程 | 0.704 3 | 0.143 7 | 0.869 3 | |
| Fe3+浓度:600×10-6 mg·L-1 | 双常数方程 | -2.603 5 | 0.389 5 | 0.917 4 | |
| 粒径>0.850 mm | 抛物线扩散方程 | 9.165 5 | 3.006 2 | 0.968 7 | |
| 粒径0.250~0.850 mm | 抛物线扩散方程 | 8.773 3 | 3.014 7 | 0.987 9 | |
| 粒径0.150~<0.250 mm | 抛物线扩散方程 | 9.767 2 | 2.618 0 | 0.991 8 | |
| 粒径<0.150 mm | 双常数方程 | 2.754 1 | 0.242 6 | 0.997 0 | |
| Tl | pH=3.0 | 抛物线扩散方程 | 2.762 1 | -0.033 8 | 0.432 0 |
| pH=4.5 | 二级动力学方程 | 0.158 7 | 0.467 2 | 0.138 0 | |
| pH=7.0 | 抛物线扩散方程 | 2.017 9 | 0.021 8 | 0.302 9 | |
| pH=9.0 | 抛物线扩散方程 | 2.757 3 | 0.014 9 | 0.383 2 | |
| Fe3+浓度:200×10-6 mg·L-1 | Elovich方程 | 5.638 9 | -0.475 4 | 0.794 6 | |
| Fe3+浓度:400×10-6 mg·L-1 | Elovich方程 | 8.459 9 | -0.793 9 | 0.867 4 | |
| Fe3+浓度:600×10-6 mg·L-1 | Elovich方程 | 10.889 2 | -1.072 1 | 0.859 0 | |
| 粒径>0.850 mm | 抛物线扩散方程 | 1.864 7 | -0.040 6 | 0.678 3 | |
| 粒径0.250~0.850 mm | 抛物线扩散方程 | 2.262 3 | -0.056 3 | 0.692 2 | |
| 粒径0.150~<0.250 mm | 双常数方程 | 0.922 6 | -0.078 9 | 0.537 4 | |
| 粒径<0.150 mm | 抛物线扩散方程 | 2.762 0 | -0.033 8 | 0.432 0 | |
表3 含砷尾矿浸出动力学方程拟合结果
Table 3 Results of linear fitting of kinetic equation of leaching of As-bearing tailings
| 元素 | 浸出环境 | 符合模型 | 参数 | ||
|---|---|---|---|---|---|
| a | b | R2 | |||
| As | pH=3.0 | 双常数方程 | 2.754 1 | 0.242 6 | 0.997 0 |
| pH=4.5 | 双常数方程 | 2.345 1 | 0.311 3 | 0.990 2 | |
| pH=7.0 | 双常数方程 | 2.418 9 | 0.297 4 | 0.993 4 | |
| pH=9.0 | 双常数方程 | 2.445 6 | 0.294 8 | 0.993 2 | |
| Fe3+浓度:200×10-6 mg·L-1 | Elovich方程 | 2.513 2 | 2.474 5 | 0.971 9 | |
| Fe3+浓度:400×10-6 mg·L-1 | 抛物线扩散方程 | 0.704 3 | 0.143 7 | 0.869 3 | |
| Fe3+浓度:600×10-6 mg·L-1 | 双常数方程 | -2.603 5 | 0.389 5 | 0.917 4 | |
| 粒径>0.850 mm | 抛物线扩散方程 | 9.165 5 | 3.006 2 | 0.968 7 | |
| 粒径0.250~0.850 mm | 抛物线扩散方程 | 8.773 3 | 3.014 7 | 0.987 9 | |
| 粒径0.150~<0.250 mm | 抛物线扩散方程 | 9.767 2 | 2.618 0 | 0.991 8 | |
| 粒径<0.150 mm | 双常数方程 | 2.754 1 | 0.242 6 | 0.997 0 | |
| Tl | pH=3.0 | 抛物线扩散方程 | 2.762 1 | -0.033 8 | 0.432 0 |
| pH=4.5 | 二级动力学方程 | 0.158 7 | 0.467 2 | 0.138 0 | |
| pH=7.0 | 抛物线扩散方程 | 2.017 9 | 0.021 8 | 0.302 9 | |
| pH=9.0 | 抛物线扩散方程 | 2.757 3 | 0.014 9 | 0.383 2 | |
| Fe3+浓度:200×10-6 mg·L-1 | Elovich方程 | 5.638 9 | -0.475 4 | 0.794 6 | |
| Fe3+浓度:400×10-6 mg·L-1 | Elovich方程 | 8.459 9 | -0.793 9 | 0.867 4 | |
| Fe3+浓度:600×10-6 mg·L-1 | Elovich方程 | 10.889 2 | -1.072 1 | 0.859 0 | |
| 粒径>0.850 mm | 抛物线扩散方程 | 1.864 7 | -0.040 6 | 0.678 3 | |
| 粒径0.250~0.850 mm | 抛物线扩散方程 | 2.262 3 | -0.056 3 | 0.692 2 | |
| 粒径0.150~<0.250 mm | 双常数方程 | 0.922 6 | -0.078 9 | 0.537 4 | |
| 粒径<0.150 mm | 抛物线扩散方程 | 2.762 0 | -0.033 8 | 0.432 0 | |

图7 不同处理组尾矿浸出前后As(a)和Tl(b)化学形态变化 F1—可交换态;F2—碳酸盐结合态;F3—铁锰氧化物结合态;F4—有机结合态;F5—残渣态。
Fig.7 Occurrences of As (a) and Tl (b) before and after leaching of tailings under different conditions. (F1: exchangeable fraction; F2: carbonate-bound fraction; F3: Fe-Mn oxide-bound fraction; F4: organic-bound fraction; F5: residual fraction)
| [1] | QIU J P, ZHAO Y L, LONG H, et al. Low-carbon binder for cemented paste backfill: flowability, strength and leaching characteristics[J]. Minerals, 2019, 9(11): 707. |
| [2] | ROUSSEL C, BRIL H, FERNANDEZ A. Arsenic speciation: involvement in evaluation of environmental impact caused by mine wastes[J]. Journal of Environmental Quality, 2000, 29(1): 182-188. |
| [3] | ASSAWINCHAROENKIJ T, HAUZENBERGER C, SUTTHIRAT C. Mineralogy and geochemistry of tailings from a gold mine in northeastern Thailand[J]. Human and Ecological Risk Assessment: An International Journal, 2017, 23(2): 364-387. |
| [4] | WU Y, ZHOU X Y, LEI M, et al. Migration and transformation of arsenic: contamination control and remediation in realgar mining areas[J]. Applied Geochemistry, 2017, 77: 44-51. |
| [5] | 马玉玲, 马杰, 陈雅丽, 等. 水铁矿及其胶体对砷的吸附与吸附形态[J]. 环境科学, 2018, 39(1):179-186. |
| [6] | LIU J, WANG J, TSANG D C W, et al. Emerging thallium pollution in China and source tracing by thallium isotopes[J]. Environmental Science and Technology, 2018, 52(21): 11977-11979. |
| [7] | LI D X, GAO Z M, ZHU Y X, et al. Photochemical reaction of Tl in aqueous solution and its environmental significance[J]. Geochemical Journal, 2005, 39(2): 113-119. |
| [8] | XIAO T F, GUHA J, BOYLE D, et al. Environmental concerns related to high thallium levels in soils and thallium uptake by plants in Southwest Guizhou, China[J]. Science of the Total Environment, 2004, 318(1/2/3): 223-244. |
| [9] | CHEN W, HUANGFU X, XIONG J, et al. Retention of thallium(I) on goethite, hematite, and manganite: quantitative insights and mechanistic study[J]. Water Research, 2022, 221: 118836. |
| [10] | WEN J C, WU Y G, LU Q, et al. Releasing characteristics and biological toxicity of the heavy metals from waste of mercury-thalliummine in Southwest Guizhou of China[J]. Bulletin of Environmental Contamination and Toxicology, 2021, 107(6): 1111-1120. |
| [11] | RAN H, GUO Z, YI L, et al. Pollution characteristics and source identification of soil metal(loid)s at an abandoned arsenic-containing mine, China[J]. Journal of Hazardous Materials, 2021, 413:125382. |
| [12] | SUN R G, GAO Y, YANG Y. Leaching of heavy metals from lead-zinc mine tailings and the subsequent migration and transformation characteristics in paddy soil[J]. Chemosphere, 2022, 291: 132792. |
| [13] | WANG P, SUN Z H, HU yuanan, et al. Leaching of heavy metals from abandoned mine tailings brought by precipitation and the associated environmental impact[J]. Science of the Total Environment, 2019, 695: 133893. |
| [14] | 陈志良, 赵述华, 钟松雄, 等. 添加稳定剂对尾矿土中砷形态及转换机制的影响[J]. 环境科学, 2016, 37(6): 2345-2352. |
| [15] | AKHAVAN A, GOLCHIN A. Estimation of arsenic leaching from Zn-Pb Mine tailings under environmental conditions[J]. Journal of Cleaner Production, 2021, 295: 126477. |
| [16] | YANG F, XIE S W, WEI C Y, et al. Arsenic characteristics in the terrestrial environment in the vicinity of the Shimen realgar mine, China[J]. Science of the Total Environment, 2018, 626: 77-86. |
| [17] | WANG X, ZHANG H, WANG L L, et al. Transformation of arsenic during realgar tailings stabilization using ferrous sulfate in a pilot-scale treatment[J]. Science of the Total Environment, 2019, 668: 32-39. |
| [18] | LARIOS R, FERNÁNDEZ-MARTÍNEZ R, SILVA V, et al. Chemical availability of arsenic and heavy metals in sediments from abandoned cinnabar mine tailings[J]. Environmental Earth Sciences, 2013, 68(2): 535-546. |
| [19] | ZHAO Z Z, ZHANG H, WANG X, et al. The mechanism of microwave-induced mineral transformation and stabilization of arsenic in realgar tailings using ferrous sulfate[J]. Chemical Engineering Journal, 2020, 393: 124732. |
| [20] | NIEVA N E, BORGNINO L, LOCATI F, et al. Mineralogical control on arsenic release during sediment-water interaction in abandoned mine wastes from the Argentina Puna[J]. Science of the Total Environment, 2016, 550: 1141-1151. |
| [21] | 贺文霄, 刘雪敏. 表生环境铊污染现状及国内外治理技术进展[J]. 复旦学报(自然科学版), 2023, 62(2):248-262. |
| [22] | 邱国良, 陈泓霖. 衡阳市湘江流域地表水铊预警监测体系探讨[J]. 供水技术, 2023, 17(3): 20-22. |
| [23] | EIGHMY T T, EUSDEN J D, KRZANOWSKI J E, et al. Comprehensive approach toward understanding element speciation and leaching behavior in municipal solid waste incineration electrostatic precipitator ash[J]. Environmental Science and Technology, 1995, 29(3): 629-646. |
| [24] | SAIKIA N, BORAH R R, KONWAR K, et al. pH dependent leachings of some trace metals and metalloid species from lead smelter slag and their fate in natural geochemical environment[J]. Groundwater for Sustainable Development, 2018, 7: 348-358. |
| [25] | COSTIS S, COUDERT L, MUELLER K, et al. Behaviour of flotation tailings from a rare earth element deposit at high salinity[J]. Journal of Environmental Management, 2021, 300: 113773. |
| [26] | XU D M, FU R, TONG Y, et al. The potential environmental risk implications of heavy metals based on their geochemical and mineralogical characteristics in the size-segregated zinc smelting slags[J]. Journal of Cleaner Production, 2021, 315: 128199. |
| [27] | VOEGELIN A, PFENNINGER N, PETRIKIS J, et al. Thallium speciation and extractability in a thallium- and arsenic-rich soil developed from mineralized carbonate rock[J]. Environmental Science and Technology, 2015, 49(9): 5390-5398. |
| [28] | WEN Q Q, YANG X, YAN X L, et al. Evaluation of arsenic mineralogy and geochemistry in gold mine-impacted matrices: speciation, transformation, and potential associated risks[J]. Journal of Environmental Management, 2022, 308: 114619. |
| [29] | KIM J Y, DAVIS A P, KIM K W. Stabilization of available arsenic in highly contaminated mine tailings using iron[J]. Environmental Science and Technology, 2003, 37(1): 189-195. |
| [30] | DRAHOTA P, KULAKOWSKI O, CULKA A, et al. Arsenic mineralogy of near-neutral soils and mining waste at the Smolotely-Líšnice historical gold district, Czech Republic[J]. Applied Geochemistry, 2018, 89: 243-254. |
| [31] | 李惠全, 胡麓华, 樊娟, 等. “十二五” 期间湖南省酸雨污染现状及成因分析研究[J]. 环境科学与管理, 2015, 40(11):57-60. |
| [32] | RIEUWERTS J S, MIGHANETARA K, BRAUNGARDT C B, et al. Geochemistry and mineralogy of arsenic in mine wastes and stream sediments in a historic metal mining area in the UK[J]. Science of the Total Environment, 2014, 472: 226-234. |
| [33] | TESSIER A, CAMPBELL P G C, BISSON M. Sequential extraction procedure for the speciation of particulate trace metals[J]. Analytical Chemistry, 1979, 51(7): 844-851. |
| [34] | MALMSTRÖM M E, GLEISNER M, HERBERT R B. Element discharge from pyritic mine tailings at limited oxygen availability in column experiments[J]. Applied Geochemistry, 2006, 21(1): 184-202. |
| [35] | 吴攀, 刘丛强, 杨元根, 等. 矿山环境中(重)金属的释放迁移地球化学及其环境效应[J]. 矿物学报, 2001, 21(2):213-218. |
| [36] | BERGER A C, BETHKE C M, KRUMHANSL J L. A process model of natural attenuation in drainage from a historic mining district[J]. Applied Geochemistry, 2000, 15(5): 655-666. |
| [37] | DONAHUE R, HENDRY M J. Geochemistry of arsenic in uranium mine mill tailings, Saskatchewan, Canada[J]. Applied Geochemistry, 2003, 18(11): 1733-1750. |
| [38] | 王云燕, 徐慧, 唐巾尧, 等. 硫化砷渣的环境稳定性与金属释放风险研究[J]. 中南大学学报(自然科学版), 2023, 54(2):548-561. |
| [39] | SAIKIA N, KATO S, KOJIMA T. Behavior of B, Cr, Se, As, Pb, Cd, and Mo present in waste leachates generated from combustion residues during the formation of ettringite[J]. Environmental Toxicology and Chemistry, 2006, 25(7): 1710-1719. |
| [40] | TURNER A, CABON A, GLEGG G A, et al. Sediment-water interactions of thallium under simulated estuarine conditions[J]. Geochimica et Cosmochimica Acta, 2010, 74(23): 6779-6787. |
| [41] | VAXEVANIDOU K, CHRISTOU C, KREMMYDAS G, et al. Role of indigenous arsenate and iron(III) respiring microorganisms in controlling the mobilization of arsenic in a contaminated soil sample[J]. Bulletin of Environmental Contamination and Toxicology, 2015, 94(3): 282-288. |
| [42] | KE W, ZENG J, ZHU F, et al. Geochemical partitioning and spatial distribution of heavy metals in soils contaminated by lead smelting[J]. Environmental Pollution, 2022, 307: 119486. |
| [43] | XIE Y Y, LU G N, YANG C F, et al. Mineralogical characteristics of sediments and heavy metal mobilization along a river watershed affected by acid mine drainage[J]. PLoS One, 2018, 13(1): e0190010. |
| [44] | DONG Y B, CHEN D N, LIN H. The behavior of heavy metal release from sulfide waste rock under microbial action and different environmental factors[J]. Environmental Science and Pollution Research, 2022, 29(50): 75293-75306. |
| [45] | ZHU W X, XIA J, YANG Y, et al. Sulfur oxidation activities of pure and mixed thermophiles and sulfur speciation in bioleaching of chalcopyrite[J]. Bioresource Technology, 2011, 102(4): 3877-3882. |
| [46] | MCKIBBEN M A. Oxidation of pyrite in low temperature acidic solutions: rate laws and surface textures[J]. Geochimica et Cosmochimica Acta, 1986, 50(7): 1509-1520. |
| [47] | RIMSTIDT J D, NEWCOMB W D. Measurement and analysis of rate data: the rate of reaction of ferric iron with pyrite[J]. Geochimica et Cosmochimica Acta, 1993, 57(9): 1919-1934. |
| [48] | NICHOLSON R V, GILLHAM R W, REARDON E J. Pyrite oxidation in carbonate-buffered solution: 1. Experimental kinetics[J]. Geochimica et Cosmochimica Acta, 1988, 52(5): 1077-1085. |
| [49] | JOHNSON R H, BLOWES D W, ROBERTSON W D, et al. The hydrogeochemistry of the nickel rim mine tailings impoundment, Sudbury, Ontario[J]. Journal of Contaminant Hydrology, 2000, 41(1/2): 49-80. |
| [50] | JOHNSON D B, HALLBERG K B. Acid mine drainage remediation options: a review[J]. Science of the Total Environment, 2005, 338(1/2): 3-14. |
| [51] | 袁婧, 吴骥子, 连斌, 等. 氧化石墨烯负载铁锰复合材料对镉砷污染土壤的钝化修复[J]. 环境科学, 2024, 45(2): 1107-1117. |
| [52] | SMEDLEY P L, KINNIBURGH D G. A review of the source, behaviour and distribution of arsenic in natural waters[J]. Applied Geochemistry, 2002, 17(5): 517-568. |
| [53] | AGUILAR-CARRILL J, HERRERA L, GUTIERREZ E J, et al. Solid-phase distribution and mobility of thallium in mining-metallurgical residues: environmental hazard implications[J]. Environmental Pollution, 2018, 243: 1833-1845. |
| [54] | BUTLER B A. Effect of pH, ionic strength, dissolved organic carbon, time, and particle size on metals release from mine drainage impacted streambed sediments[J]. Water Research, 2009, 43(5): 1392-1402. |
| [55] | CHEN T, YAN Z A, XU D M, et al. Current situation and forecast of environmental risks of a typical lead-zinc sulfide tailings impoundment based on its geochemical characteristics[J]. Journal of Environmental Sciences, 2020, 93: 120-128. |
| [1] | 鹿帅, 苏小四, 冯晓语, 孙超. 河水入渗过程中近岸带地下水中砷的形成与影响因素研究[J]. 地学前缘, 2022, 29(4): 455-467. |
| [2] | 张玉婷, 段丽琴, 宋金明, 张乃星, 尹美玲, 李学刚, 袁华茂. 长江口沉积物-水界面砷的迁移转化机制与微生物调控分析[J]. 地学前缘, 2022, 29(4): 144-155. |
| [3] | 孙英, 周金龙, 杨方源, 纪媛媛, 曾妍妍. 塔里木盆地南缘绿洲带地下水砷氟碘分布及共富集成因[J]. 地学前缘, 2022, 29(3): 99-114. |
| [4] | 吕晓立, 郑跃军, 韩占涛, 李海军, 杨明楠, 张若琳, 刘丹丹. 城镇化进程中珠江三角洲地区浅层地下水中砷分布特征及成因[J]. 地学前缘, 2022, 29(3): 88-98. |
| [5] | 郭华明, 高志鹏, 修伟. 地下水典型氧化还原敏感组分迁移转化的研究热点和趋势[J]. 地学前缘, 2022, 29(3): 64-75. |
| [6] | 张耀强, 胡冰冰, 谢世伟, 袁松虎. 地下水曝气-砂滤处理产生的废弃石英砂中铁砷含量与形态分布[J]. 地学前缘, 2021, 28(5): 208-214. |
| [7] | Jinru Lin, Ning Chen, Yuanming Pan. 砷在石膏中固定机制:掺杂态和表面吸附沉淀态以及其在砷污染控制中的作用[J]. 地学前缘, 2020, 27(5): 227-237. |
| [8] | 岳昌盛,都慧丽,彭犇,崔岩山. 利用体外方法评估不同淋洗剂对土壤中砷的淋洗效果及其健康风险 [J]. 地学前缘, 2019, 26(6): 42-48. |
| [9] | 梁梦钰,郭华明,李晓萌,王振,修伟. 贵德盆地三河流域高砷地下水中溶解性有机物三维荧光特性及其指示意义[J]. 地学前缘, 2019, 26(3): 243-254. |
| [10] | 曹永生,郭华明,倪萍,李山羊,郭琦,韩双宝,李付兰. 沉积物地球化学特征和土地利用方式对地下水砷行为的影响[J]. 地学前缘, 2017, 24(2): 274-285. |
| [11] | 甘义群, 王焰新, 段艳华, 邓娅敏, 郭欣欣. 江汉平原高砷地下水监测场砷的动态变化特征分析[J]. 地学前缘, 2014, 21(4): 37-49. |
| [12] | 汤洁, 卞建民, 李昭阳, 张楠, 梁爽, 邴志武. 吉林省饮水型砷中毒区地下水砷的分布规律与成因研究[J]. 地学前缘, 2014, 21(4): 30-36. |
| [13] | 郭华明, 倪萍, 贾永锋, 郭琦, 姜玉肖. 原生高砷地下水的类型、化学特征及成因[J]. 地学前缘, 2014, 21(4): 1-12. |
| [14] | 谢越, 周立祥. 酸性环境下生物成因施氏矿物稳定性研究[J]. 地学前缘, 2011, 18(5): 310-318. |
| [15] | 周立祥. 酸性矿山废水中生物成因次生高铁矿物的形成及环境工程意义[J]. 地学前缘, 2008, 15(6): 74-82. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||