

地学前缘 ›› 2020, Vol. 27 ›› Issue (3): 42-67.DOI: 10.13745/j.esf.sf.2020.5.56
• “非传统稳定同位素:分析方法、示踪机理和主要应用”主题专辑 • 上一篇 下一篇
收稿日期:2020-04-14
修回日期:2020-04-19
出版日期:2020-05-20
发布日期:2020-05-20
通信作者:
王瑞
作者简介:周秋石(1999—),男,本科生,研究方向为矿床地球化学。E-mail: zhouqiushi121@gmail.com
基金资助:Received:2020-04-14
Revised:2020-04-19
Online:2020-05-20
Published:2020-05-20
Contact:
WANG Rui
摘要:
作为最具代表性的、地球丰度最高的卤素,氯元素因其独特的性质和各大储库中的可观分布而受到重视。氯元素具有显著的亲硫性和挥发性(不相容性),这些性质也影响着其地球化学行为以及在地球中的分布。氯含有两种稳定同位素,分别是35Cl和37Cl,同位素丰度分别为75.76%和24.24%,Cl同位素组成也常以δ37Cl值报道;目前对Cl同位素进行分析的最为传统也是最主要的方法是IRMS(同位素比值质谱法),尽管存在分析速度慢、对样品质量要求大等缺陷,但因其相对更高的精度而被广泛应用于现代氯稳定同位素研究。其他常见的分析方法还有TIMS,SIMS,LA-ICP-MS,在Cl同位素研究领域尚在实验探索阶段。Cl同位素标准在目前也已有广泛接受的统一国际标准,即Kaufmann所提出的平均海洋Cl同位素标准(SMOC),海水Cl同位素组成在长久演化中已经相对稳定,取样也简单方便,在实验室分析中也能产生优良的再现性。氯在地球上的储库可在宏观上分为地幔、陆壳与洋壳、海洋。地幔以其较大的体量无疑是氯元素主要储库之一,但受限于现有观测技术,各研究通过各种方法观测到的氯含量有较大出入;关于地幔的Cl同位素分布也并不明确,各种如俯冲带流体输入等深部过程都可能是同位素异质性的原因,许多研究观测到的地幔流体的Cl同位素有正也有负。陆壳可下分为沉积物及其孔隙水、蒸发岩和硅质岩石圈,氯因其亲水性和化学沉积性质则主要集中于沉积物孔隙水和蒸发岩中,而硅质岩石圈中的氯则相对较少;沉积物孔隙水常被观测到极低的δ37Cl值,扩散和离子淋滤都是对其可能的解释,但对其完全做出解释的机理还有待继续研究;蒸发岩中的δ37Cl值常因其氯盐种类而有所不同,也已有诸多实验给出制约;硅质岩石圈虽含氯较少,但出露于地表的岩株或岩脉中观测到的磷灰石中的Cl同位素有明显的随岩性变化的规律性,对于热液交代历史与热液成分都有很好的限制作用。洋壳中的氯也主要分布在洋壳沉积物及其孔隙水、蒸发岩,以及蚀变洋壳(以角闪石、蛇纹石等蚀变矿物为代表)中。蚀变矿物常被观察到较高的δ37Cl值,一些研究认为可能和与氯结合的金属阳离子氧化状态有关,该高δ37Cl值也常被用来解释俯冲板片输入地幔流体的高δ37Cl值。海洋也是巨大的氯元素储库,其同位素组成稳定性也已经得到证明。除以上储库以外,Cl同位素还应用于对陨石、月球和大气圈的研究。Cl同位素的分馏机制也同其他许多同位素一样可大体分为平衡分馏和动力学分馏,平衡分馏主要包括由与氯结合的金属阳离子的价态高低或氯自身价态的高低决定的分馏,动力学分馏则包括扩散和离子淋滤以及与地球岩浆去气、月球岩浆去气有关的动力学分馏。总体而言,氯元素及与其结合的金属离子的氧化状态、轻重同位素逃逸倾向、扩散系数的差异是决定分馏行为的两种机理。从氯的两种稳定同位素自大约100年前被发现起到现在,与氯稳定同位素相关的地球化学方法已经在各地质学科分支中得到广泛应用。从利用Cl同位素解释地层水来源的水文地质、示踪污染物来源的环境地质,到利用Cl同位素证据支撑矿床形成机制研究的矿床学以及参与解释地球演化过程的行星地质等领域,都有Cl同位素应用研究的尝试与探索。但受限于对实际地质过程的认知,仍然具有许多前沿问题阻碍Cl同位素研究的进展,如太阳系早期过程如何决定了地球现今的氯元素含量、地球中的挥发分经历了怎样的演化过程才达到至今状态、现今地球挥发分的循环过程又具体如何、俯冲过程中氯在俯冲带与地幔之间的流通过程具体如何等。
中图分类号:
周秋石, 王瑞. 氯同位素地球化学研究进展[J]. 地学前缘, 2020, 27(3): 42-67.
ZHOU Qiushi, WANG Rui. Advances in chlorine isotope geochemistry[J]. Earth Science Frontiers, 2020, 27(3): 42-67.
| 储库 | 储库质量/kg | 占地球质量比例/% | Cl质量分数/10-6 | Cl质量/kg | Cl质量占 总质量比例/% |
|---|---|---|---|---|---|
| 大气圈 | 5.1×1018 | 0.000 1 | 0.001 6 | 8.16×109 | <10-8 |
| 海洋 | 1.4×1021 | 0.023 | 19 353 | 2.71×1019 | 17.8 |
| 蒸发岩 | 3.12×1019 | 0.033 | 606 838 449 509 | 1.89×1019 (1.4×1019) | 12.4 |
| 沉积物(含孔隙水) | 1.97×1021 | 0.033 | 19 206 | 3.78×1019 | 24.8 |
| 陆壳 | 1.08×1022 | 0.18 | 210 | 2.27×1018 | 1.5 |
| 洋壳 | 4.8×1021 | 0.08 | 48 | 2.30×1017 | 0.2 |
| 地幔 | 4.01×1024 | 67.07 | 16 11 | 6.59×1019 (4.6×1019) | 43.3 |
| 地核 | 1.95×1024 | 32.61 | |||
| 地球 | 5.98×1024 | 100.03 | 17.39 25.42 | (1.04×1020) 1.52×1020 | 100.0 |
表1 氯在地球各大储库中的含量
Table 1 Chlorine in the various reservoirs on the Earth
| 储库 | 储库质量/kg | 占地球质量比例/% | Cl质量分数/10-6 | Cl质量/kg | Cl质量占 总质量比例/% |
|---|---|---|---|---|---|
| 大气圈 | 5.1×1018 | 0.000 1 | 0.001 6 | 8.16×109 | <10-8 |
| 海洋 | 1.4×1021 | 0.023 | 19 353 | 2.71×1019 | 17.8 |
| 蒸发岩 | 3.12×1019 | 0.033 | 606 838 449 509 | 1.89×1019 (1.4×1019) | 12.4 |
| 沉积物(含孔隙水) | 1.97×1021 | 0.033 | 19 206 | 3.78×1019 | 24.8 |
| 陆壳 | 1.08×1022 | 0.18 | 210 | 2.27×1018 | 1.5 |
| 洋壳 | 4.8×1021 | 0.08 | 48 | 2.30×1017 | 0.2 |
| 地幔 | 4.01×1024 | 67.07 | 16 11 | 6.59×1019 (4.6×1019) | 43.3 |
| 地核 | 1.95×1024 | 32.61 | |||
| 地球 | 5.98×1024 | 100.03 | 17.39 25.42 | (1.04×1020) 1.52×1020 | 100.0 |
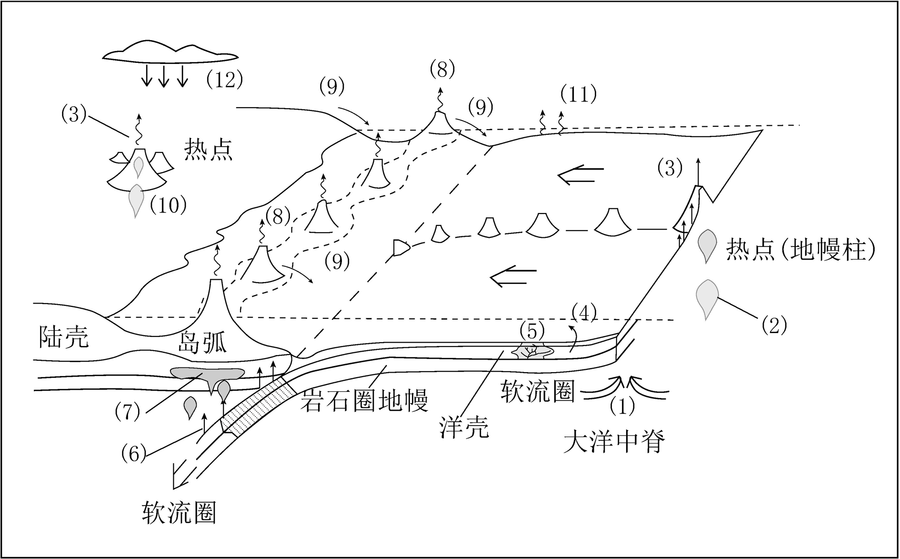
图1 与热点及板块活动相关的Cl元素地质循环(据文献[16]修改) (1)—Cl从软流圈经由洋中脊火山活动暂时储存于洋壳;(2)—Cl从地幔经由热点或地幔柱成因的火山向上运输;(3)—Cl由地表的地幔柱火山喷出;(4)—低温条件下海水与洋壳在水岩反应中的Cl交换;(5)—热液循环的运移作用,以及与之相关的洋壳交代蚀变带来的Cl运移;(6)—岩石圈俯冲过程中由脱水和变质引起的Cl损失;(7)—Cl从软流圈经由岛弧火山活动暂存于岛弧火山;(8)—Cl由岛弧火山喷出;(9)—Cl在地表风化的作用下运输;(10)—Cl在地幔柱火山活动下向上运输;(11)—海水蒸发;(12)—含Cl雨水降至地表参与风化与地下水循环。
Fig.1 Diagram showing the geological cycle of Cl associated with hotspot volcanism and plate motion. Modified after [16].
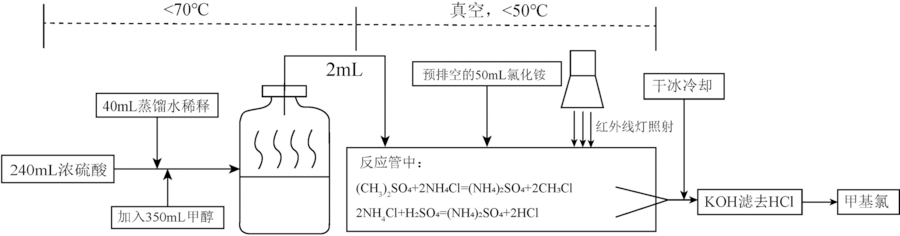
图2 由氯化铵制备氯甲烷流程简化图(据文献[57],图中所涉仪器非真实模拟)
Fig.2 Diagrammatic sketch of the preparation procedure of chloromethane from ammonium chloride. Graphed according to [57]. The equipment is not real.
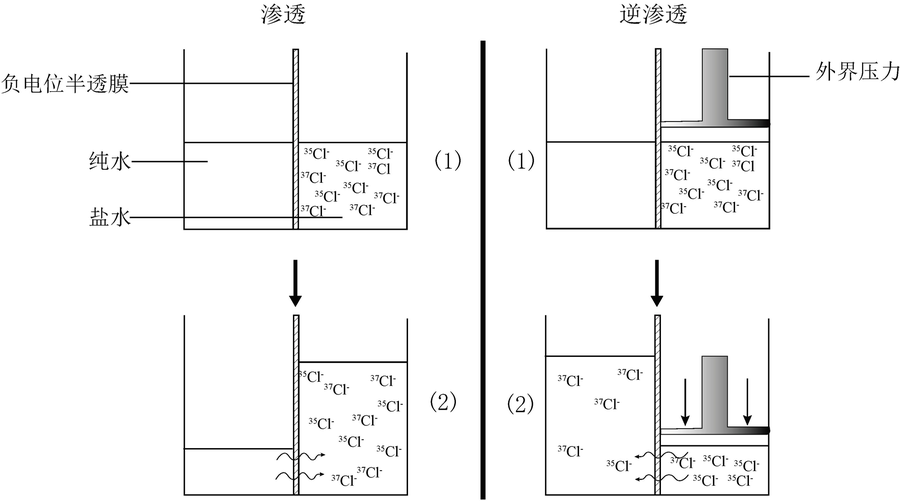
图4 离子淋滤Cl同位素分馏原理示意图。离子淋滤被认为可以比作逆向渗透过程,即“逆渗透”(据文献[42]修改)
Fig.4 Diagram showing the Cl isotope fractionation mechanism of ion filtration. Modified after [42].
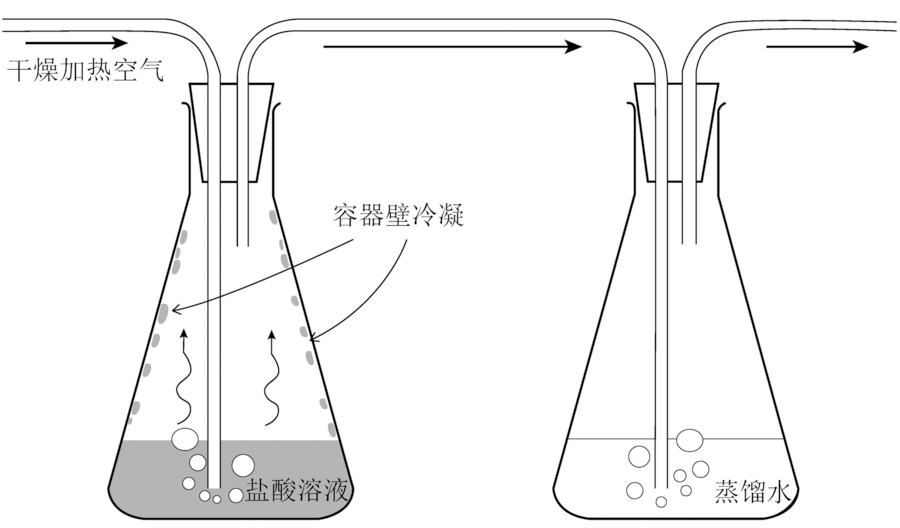
图5 动力学Cl同位素分馏“流通实验”实验设计(据文献[84]修改)
Fig.5 Schematic illustration of experimental design for Cl isotope kinetic flow-through experiments. Modified after [84].
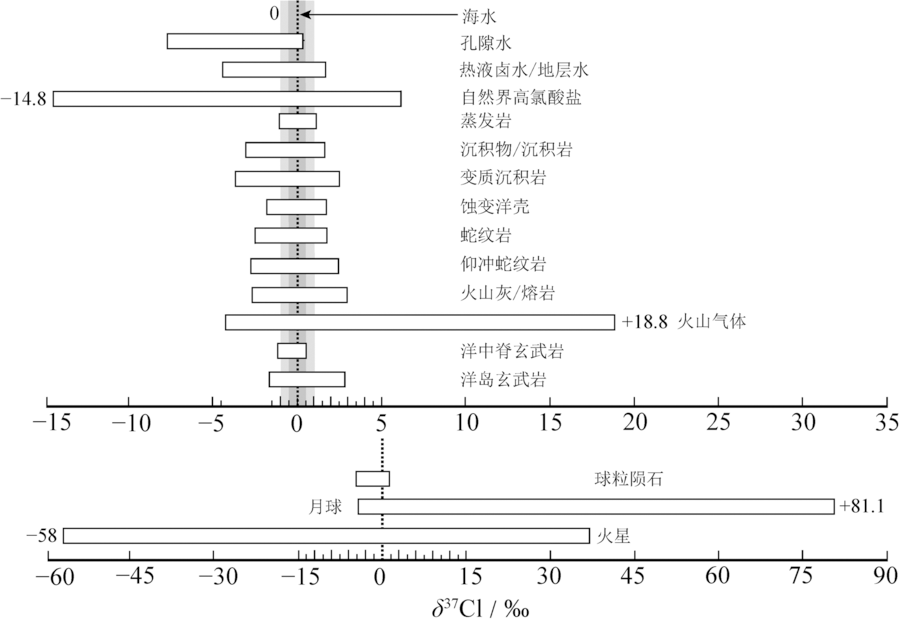
图6 Cl同位素在不同地质储库中的组成分布(据文献[99]修改) δ37Cl范围基于平均海水氯标准(SMOC)得出。两个灰度带分别代表-1.0‰~+1.0‰和-0.5‰~+0.5‰的组成范围。该图中,地球储库中δ37Cl最高值来自[98]测定火山气体得出的+18.8‰,最低值来自[101]测定微生物环境下的高氯酸盐得出的-14.8‰;天外储库中δ37Cl最高值来自[102]在月球磷灰石中测得的+81.1‰,最低值来自[103]在火星高氯酸盐中测得的-58‰。孔隙水数据来源于[71,95,104];热液卤水/地层水数据来源于[11,86,96,105-110];自然界高氯酸盐数据来源于[101,111-112];蒸发岩数据来源于[45,85-87,108];沉积物/沉积岩数据来源于[20,98,113];变质沉积岩数据来源于[75,113];蚀变洋壳、蛇纹岩数据来源于[19-20,71,98,114-115];仰冲蛇纹岩数据来源于[19,116];火山灰/熔岩数据来源于[20-21,98,100];火山气体数据来源于[20,98,100];洋中脊玄武岩数据来源于[46];洋岛玄武岩数据来源于[75];球粒陨石数据来源于[46];月球数据来源于[76,102,117-118];火星数据来源于[103,119]。
Fig.6 Chlorine isotope variability in different terrestrial and extraterrestrial reservoirs. Modified after [99].

图7 伊豆-小笠原-马里亚纳(IBM)俯冲带岛弧火山体系的δ37Cl值来源与去向图(据文献[20]修改,岛弧岩浆的δ37Cl值在该研究中被认为继承于俯冲带流体)
Fig.7 Schematic diagram showing fluid sources in subducting slab, outputs across the arc, and their average δ37Cl values. Modified after [20]. The δ37Cl value of island arc magma is considered to heritage from subduction fluid.
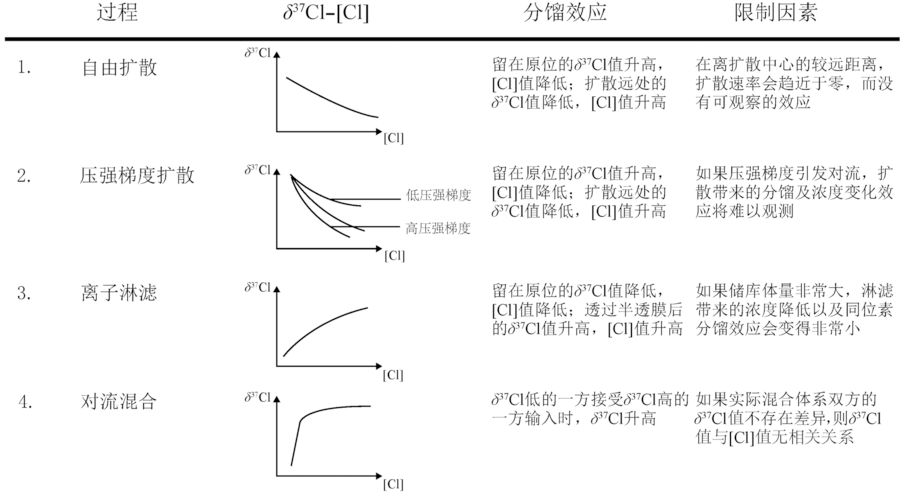
图8 扩散、离子淋滤与对流混合对Cl同位素及氯含量的效应(据文献[107]修改)
Fig.8 Chlorine isotope effects of diffusion-related processes, ion filtration and advective mixing. Modified after [107].
| [1] |
SUN S S. Chemical composition and origin of the Earth's primitive mantle[J]. Geochimica et Cosmochimica Acta, 1982, 46(2):179-192.
DOI URL |
| [2] |
UREY H C. The origin and development of the earth and other terrestrial planets[J]. Geochimica et Cosmochimica Acta, 1951, 1(4/5/6):209-277.
DOI URL |
| [3] | SHARP Z D, DRAPER D S. The chlorine abundance of Earth: implications for a habitable planet[J]. Earth and Planetary Science Letters, 2013, 369:71-77. |
| [4] |
ALLEGRE C, MANHES G, LEWIN É. Chemical composition of the Earth and the volatility control on planetary genetics[J]. Earth and Planetary Science Letters, 2001, 185(1/2):49-69.
DOI URL |
| [5] |
MCDONOUGH W F, SUN S S. The composition of the Earth[J]. Chemical Geology, 1995, 120(3/4):223-253.
DOI URL |
| [6] | EGGENKAMP H. The geochemistry of stable chlorine and bromine isotopes[M]. Berlin: Springer, 2014: 8-10, 16, 31-37. |
| [7] | STACEY F D, DAVIS P M. Physics of the Earth[M]. Cambridge: Cambridge University Press, 2008. |
| [8] | RAHN K A. The chemical composition of the atmospheric aerosol[M]. Kingston: Graduate School of Oceanography, University of Rhode Island, 1976. |
| [9] | PYTKOWICZ R M, KESTER D R. The physical chemistry of seawater[J]. Annual Review Oceanographic Marine Biology, 1971, 9:11-60. |
| [10] |
HAY W W, MIGDISOV A, BALUKHOVSKY A N, et al. Evaporites and the salinity of the ocean during the Phanerozoic: Implications for climate, ocean circulation and life[J]. Palaeogeography, Palaeoclimatology, Palaeoecology, 2006, 240(1/2):3-46.
DOI URL |
| [11] |
EGGENKAMP H G M, MIDDELBURG J J, KREULEN R. Preferential diffusion of35Cl relative to37Cl in sediments of Kau Bay, Halmahera, Indonesia[J]. Chemical Geology, 1994, 116(3/4):317-325.
DOI URL |
| [12] |
LAND L S. The role of saline formation water in crustal cycling[J]. Aquatic Geochemistry, 1995, 1(2):137-145.
DOI URL |
| [13] |
KNAUTH L P. Salinity history of the Earth's early ocean[J]. Nature, 1998, 395(6702):554-555.
DOI URL |
| [14] | GAST P W. The chemical composition of the earth, the moon, and chondritic meteorites[M]// The nature of the solid Earth. New York: McGraw-Hill, 1972: 19-40. |
| [15] | TUREKIAN K K. Geochemical distribution of elements[J]. Encyclopedia of Science and Technology, 1977, 4:627-630. |
| [16] |
SCHILLING J G, UNNI C K, BENDER M L. Origin of chlorine and bromine in the oceans[J]. Nature, 1978, 273(5664):631-636.
DOI URL |
| [17] | DUBOIS J L, OJHA S. Production of dioxygen in the dark: dismutases of oxyanions[M]// Sustaining life on planet earth: metalloenzymes mastering dioxygen and other chewy gases. Cham: Springer, 2015: 45-87. |
| [18] |
RAJAGOPALAN S, ANDERSON T A, FAHLQUIST L, et al. Widespread presence of naturally occurring perchlorate in high plains of Texas and New Mexico[J]. Environmental Science & Technology, 2006, 40(10):3156-3162.
DOI URL |
| [19] |
BONIFACIE M, BUSIGNY V, MéVEL C, et al. Chlorine isotopic composition in seafloor serpentinites and high-pressure metaperidotites. Insights into oceanic serpentinization and subduction processes[J]. Geochimica et Cosmochimica Acta, 2008, 72(1):126-139.
DOI URL |
| [20] |
BARNES J D, SHARP Z D, FISCHER T P. Chlorine isotope variations across the Izu-Bonin-Mariana arc[J]. Geology, 2008, 36(11):883-886.
DOI URL |
| [21] |
CHIARADIA M, BARNES J D, CADET-VOISIN S. Chlorine stable isotope variations across the Quaternary volcanic arc of Ecuador[J]. Earth and Planetary Science Letters, 2014, 396:22-33.
DOI URL |
| [22] |
ZHANG C, HOLTZ F, MA C, et al. Tracing the evolution and distribution of F and Cl in plutonic systems from volatile-bearing minerals: a case study from the Liujiawa pluton (Dabie orogen, China)[J]. Contributions to Mineralogy and Petrology, 2012, 164(5):859-879.
DOI URL |
| [23] |
KONZETT J, RHEDE D, FROST D J. The high p-T stability of apatite and Cl partitioning between apatite and hydrous potassic phases in peridotite: an experimental study to 19 GPa with implications for the transport of P, Cl and K in the upper mantle[J]. Contributions to Mineralogy and Petrology, 2012, 163(2):277-296.
DOI URL |
| [24] | MUNOZ J L. F-OH and Cl-OH exchange in micas with applications to hydrothermal ore deposits[J]. Micas, 2018, 13:469. |
| [25] |
KURODA P K, SANDELL E B. Chlorine in igneous rocks: some aspects of the geochemistry of chlorine[J]. Geological Society of America Bulletin, 1953, 64(8):879-896.
DOI URL |
| [26] |
YOSHIDA M, TAKAHASHI K, YONEHARA N, et al. The fluorine, chlorine, bromine, and iodine contents of volcanic rocks in Japan[J]. Bulletin of the Chemical Society of Japan, 1971, 44(7):1844-1850.
DOI URL |
| [27] |
ANDERSON A T. Chlorine, sulfur, and water in magmas and oceans[J]. Geological Society of America Bulletin, 1974, 85(9):1485-1492.
DOI URL |
| [28] |
JOHNSTON D A. Volcanic contribution of chlorine to the stratosphere: more significant to ozone than previously estimated?[J]. Science, 1980, 209(4455):491-493.
DOI URL |
| [29] |
MUNGALL J E, BRENAN J M. Experimental evidence for the chalcophile behavior of the halogens[J]. The Canadian Mineralogist, 2003, 41(1):207-220.
DOI URL |
| [30] |
STEENSTRA E S, VAN HAASTER F, VAN MULLIGEN R, et al. An experimental assessment of the chalcophile behavior of F, Cl, Br and I: implications for the fate of halogens during planetary accretion and the formation of magmatic ore deposits[J]. Geochimica et Cosmochimica Acta, 2020, 273:275-290.
DOI URL |
| [31] | JAGO B C, MORRISON G G, LITTLE T L. Metal zonation patterns and microtextural and micromineralogical evidence for alkali-and halogen-rich fluids in the genesis of the Victor Deep and McCreedy East footwall copper orebodies, Sudbury Igneous Complex[J]. Sudbury Igneous Complex: Ontario Geological Survey Special, 1994, 5:65-75. |
| [32] |
WILLMORE C C, BOUDREAU A E, KRUGER F J. The halogen geochemistry of the Bushveld Complex, Republic of South Africa: implications for chalcophile element distribution in the lower and critical zones[J]. Journal of Petrology, 2000, 41(10):1517-1539.
DOI URL |
| [33] | HANLEY J J. The distribution of the halogens in Sudbury Breccia matrix as pathfinder elements for footwall copper-PGE mineralization at the Fraser copper zone, Barnet main copper zone, and surrounding margin of the Sudbury igneous complex, Onaping-Levack area, Ontario, Canada[D]. Toronto: University of Toronto, 2003. |
| [34] |
CANDELA P A, HOLLAND H D. The partitioning of copper and molybdenum between silicate melts and aqueous fluids[J]. Geochimica et Cosmochimica Acta, 1984, 48(2):373-380.
DOI URL |
| [35] |
HACK A C, MAVROGENES J A. A synthetic fluid inclusion study of copper solubility in hydrothermal brines from 525 to 725 ℃ and 0.3 to 1.7 GPa[J]. Geochimica et Cosmochimica Acta, 2006, 70(15):3970-3985.
DOI URL |
| [36] |
RUBEY W W. Geologic history of sea water: an attempt to state the problem[J]. Geological Society of America Bulletin, 1951, 62(9):1111-1148.
DOI URL |
| [37] |
ANDERSON A T. Some basaltic and andesitic gases[J]. Reviews of Geophysics, 1975, 13(1):37-55.
DOI URL |
| [38] |
ITO E, HARRIS D M, ANDERSON A TJr. Alteration of oceanic crust and geologic cycling of chlorine and water[J]. Geochimica et Cosmochimica Acta, 1983, 47(9):1613-1624.
DOI URL |
| [39] |
SYMONDS R B, ROSE W I, REED M H. Contribution of Cl- and F- bearing gases to the atmosphere by volcanoes[J]. Nature, 1988, 334(6181):415-418.
DOI URL |
| [40] | JARRARD R D. Subduction fluxes of water, carbon dioxide, chlorine, and potassium[J]. Geochemistry, Geophysics, Geosystems, 2003, 4(5):8905. |
| [41] |
EGGENKAMP H G M, LOUVAT P, AGRINIER P, et al. The bromine and chlorine isotope composition of primary halite deposits and their significance for the secular isotope composition of seawater[J]. Geochimica et Cosmochimica Acta, 2019, 264:13-29.
DOI URL |
| [42] | KAUFMANN R S. Chlorine in ground water: Stable isotope distribution[D]. Tucson: University of Arizona, 1984. |
| [43] |
GODON A, JENDRZEJEWSKI N, EGGENKAMP H G M, et al. A cross-calibration of chlorine isotopic measurements and suitability of seawater as the international reference material[J]. Chemical Geology, 2004, 207(1/2):1-12.
DOI URL |
| [44] |
ZHOU A, GAN Y, LIU C, et al. An online method to determine chlorine stable isotope composition by continuous flow isotope ratio mass spectrometry (CF-IRMS) coupled with a Gasbench II[J]. Journal of Central South University, 2013, 20(1):193-198.
DOI URL |
| [45] |
EASTOE C J, PERYT T M, PETRYCHENKO Y, et al. Stable chlorine isotopes in Phanerozoic evaporites[J]. Applied Geochemistry, 2007, 22(3):575-588.
DOI URL |
| [46] |
SHARP Z D, BARNES J D, BREARLEY A J, et al. Chlorine isotope homogeneity of the mantle, crust and carbonaceous chondrites[J]. Nature, 2007, 446(7139):1062-1065.
DOI URL |
| [47] | SHIRODKAR P V, XIAO Y K, HAI L. Boron and chlorine isotopic signatures of seawater in the Central Indian Ridge[J]. Current Science, 2003, 85(3):313-320. |
| [48] |
XIAO Y K, YINMING Z, QINGZHONG W, et al. A secondary isotopic reference material of chlorine from selected seawater[J]. Chemical Geology, 2002, 182(2/3/4):655-661.
DOI URL |
| [49] |
HAN J L, HUSSAIN S A, HAN F Q. Stable chlorine isotopes in saline springs from the Nangqen basin, Qinghai-Tibet Plateau: brine genesis and evolution[J]. Journal of Earth System Science, 2019, 128(8):206.
DOI URL |
| [50] |
LI Q, ZHANG X, FAN Q, et al. Influence of non-marine fluid inputs on potash deposits in northeastern Thailand: evidence from δ37Cl value and Br/Cl ratio of halite[J]. Carbonates and Evaporites, 2020, 35(1):1-10.
DOI URL |
| [51] |
WEI H Z, JIANG S Y, XIAO Y K, et al. Precise determination of the absolute isotopic abundance ratio and the atomic weight of chlorine in three international reference materials by the positive thermal ionization mass spectrometer-Cs2Cl+-graphite method[J]. Analytical Chemistry, 2012, 84(23):10350-10358.
DOI URL |
| [52] | HOEFS J. Stable isotope geochemistry[M]. Berlin: Springer, 2009. |
| [53] |
ASTON F W. A positive ray spectrograph[J]. The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science, 1919, 38(228):707-714.
DOI URL |
| [54] |
MADORSKY S L, STRAUS S. Concentration of isotopes of chlorine by the counter-current electromigration method[J]. Journal of Research of the National Bureau of Standards, 1947, 38:185-189.
PMID |
| [55] |
HOERING T C, PARKER P L. The geochemistry of the stable isotopes of chlorine[J]. Geochimica et Cosmochimica Acta, 1961, 23(3/4):186-199.
DOI URL |
| [56] |
MAGENHEIM A J, SPIVACK A J, VOLPE C, et al. Precise determination of stable chlorine isotopic ratios in low-concentration natural samples[J]. Geochimica et Cosmochimica Acta, 1994, 58(14):3117-3121.
DOI URL |
| [57] | BLATT A H. Organic Synbook, Collective Volume 2[M]. New York: Wiley, 1943: 251. |
| [58] | LANGVAD T. Separation of chlorine isotopes by ion-exchange chromatography[J]. Acta Chemica Scandinavica (Denmark): Series A and Series B, 1954, 8:526-527. |
| [59] | EGGENKAMP H. δ37Cl: the geochemistry of chlorine isotopes[J]. Geologica Ultraiectina, 1994, 116:1-150. |
| [60] |
HOLT B D, STURCHIO N C, ABRAJANO T A, et al. Conversion of chlorinated volatile organic compounds to carbon dioxide and methyl chloride for isotopic analysis of carbon and chlorine[J]. Analytical Chemistry, 1997, 69(14):2727-2733.
DOI URL |
| [61] |
FUJITANI T, YAMASHITA K, NUMATA M, et al. Measurement of chlorine stable isotopic composition by negative thermal ionization mass spectrometry using total evaporation technique[J]. Geochemical Journal, 2010, 44(3):241-246.
DOI URL |
| [62] |
XIAO Y K, ZHANG C G. High precision isotopic measurement of chlorine by thermal ionization mass spectrometry of the Cs2Cl+ ion[J]. International Journal of Mass Spectrometry and Ion Processes, 1992, 116(3):183-192.
DOI URL |
| [63] |
BANKS D A, GREEN R, CLIFF R A, et al. Chlorine isotopes in fluid inclusions: determination of the origins of salinity in magmatic fluids[J]. Geochimica et Cosmochimica Acta, 2000, 64(10):1785-1789.
DOI URL |
| [64] |
MAGENHEIM A J, SPIVACK A J, MICHAEL P J, et al. Chlorine stable isotope composition of the oceanic crust: Implications for Earth's distribution of chlorine[J]. Earth and Planetary Science Letters, 1995, 131(3/4):427-432.
DOI URL |
| [65] |
SHIRODKAR P V, XIAO Y K, SARKAR A, et al. Influence of air-sea fluxes on chlorine isotopic composition of ocean water: implications for constancy in δ37Cl: A statistical inference[J]. Environment International, 2006, 32(2):235-239.
DOI URL |
| [66] | NAKAMURA N, NYQUIST L E, REESE Y, et al. Stable chlorine isotopes and elemental chlorine by thermal ionization mass spectrometry and ion chromatography: Martian meteorites, carbonaceous chondrites and standard rocks[C]// Proceedings of the 42nd Lunar and Planetary Science Conference, 2011, 42:2513. |
| [67] |
MAGENHEIM A J, SPIVACK A J, VOLPE C, et al. Precise determination of stable chlorine isotopic ratios in low-concentration natural samples[J]. Geochimica et Cosmochimica Acta, 1994, 58(14):3117-3121.
DOI URL |
| [68] |
XIAO Y K, ZHOU Y M, LIU W G. Precise measurement of chlorine isotopes based on Cs2Cl2 by thermal ionization mass spectrometry[J]. Analytical Letters, 1995, 28(7):1295-1304.
DOI URL |
| [69] | STEWART MA. Geochemistry of dikes and lavas from Hess Deep: implications for crustal construction processes beneath Mid-Ocean Ridges and the stable-chlorine isotope geochemistry of Mid-Ocean Ridge Basalt glasses[D]. Durham: Duke University, 2002. |
| [70] |
ROSENBAUM J M, CLIFF R A, COLEMAN M L. Chlorine stable isotopes: a comparison of dual inlet and thermal ionization mass spectrometric measurements[J]. Analytical Chemistry, 2000, 72(10):2261-2264.
DOI URL |
| [71] |
BONIFACIE M, JENDRZEJEWSKI N, AGRINIER P, et al. Pyrohydrolysis-IRMS determination of silicate chlorine stable isotope compositions: application to oceanic crust and meteorite samples[J]. Chemical Geology, 2007, 242(1/2):187-201.
DOI URL |
| [72] |
SHARP Z D, BARNES J D. Comment to “Chlorine stable isotopes and halogen concentrations in convergent margins with implications for the Cl isotopes cycle in the ocean” by Wei et al. A review of the Cl isotope composition of serpentinites and the global chlorine cycle[J]. Earth and Planetary Science Letters, 2008, 274(3/4):531-534.
DOI URL |
| [73] |
MANZINI M, BOUVIER A S, BARNES J D, et al. SIMS chlorine isotope analyses in melt inclusions from arc settings[J]. Chemical Geology, 2017, 449:112-122.
DOI URL |
| [74] |
LAYNE G D, GODON A, WEBSTER J D, et al. Secondary ion mass spectrometry for the determination of δ37Cl: Part I. Ion microprobe analysis of glasses and fluids[J]. Chemical Geology, 2004, 207(3/4):277-289.
DOI URL |
| [75] |
JOHN T, LAYNE G D, HAASE K M, et al. Chlorine isotope evidence for crustal recycling into the Earth's mantle[J]. Earth and Planetary Science Letters, 2010, 298(1/2):175-182.
DOI URL |
| [76] |
SHARP Z D, SHEARER C K, MCKEEGAN K D, et al. The chlorine isotope composition of the Moon and implications for an anhydrous mantle[J]. Science, 2010, 329(5995):1050-1053.
DOI URL |
| [77] | FLORES E M M, MELLO P A, KRZYZANIAK S R, et al. Challenges and trends for halogen determination by inductively coupled plasma mass spectrometry: a review[J]. Rapid Communications in Mass Spectrometry, 2020: e8727. |
| [78] |
FIETZKE J, FRISCHE M, HANSTEEN T H, et al. A simplified procedure for the determination of stable chlorine isotope ratios (δ37Cl) using LA-MC-ICP-MS[J]. Journal of Analytical Atomic Spectrometry, 2008, 23(5):769-772.
DOI URL |
| [79] |
VAN ACKER M R M D, SHAHAR A, YOUNG E D, et al. GC/multiple collector-ICPMS method for chlorine stable isotope analysis of chlorinated aliphatic hydrocarbons[J]. Analytical Chemistry, 2006, 78(13):4663-4667.
DOI URL |
| [80] | UREY H C. The thermodynamic properties of isotopic substances[J]. Journal of the Chemical Society (Resumed), 1947, 0:562-581. |
| [81] |
SCHAUBLE E A, ROSSMAN G R, TAYLOR JR H P. Theoretical estimates of equilibrium chlorine-isotope fractionations[J]. Geochimica et Cosmochimica Acta, 2003, 67(17):3267-3281.
DOI URL |
| [82] | CISNEROS M. An experimental calibration of chlorine isotope fractionation between amphibole and fluid at 700 ℃ and 0.2 GPa[D]. Austin: University of Texas, 2013. |
| [83] |
AEPPLI C, BASTVIKEN D, ANDERSSON P, et al. Chlorine isotope effects and composition of naturally produced organochlorines from chloroperoxidases, flavin-dependent halogen gases, and in forest soil[J]. Environmental Science & Technology, 2013, 47(13):6864-6871.
DOI URL |
| [84] |
SHARP Z D, BARNES J D, FISCHER T P, et al. An experimental determination of chlorine isotope fractionation in acid systems and applications to volcanic fumaroles[J]. Geochimica et Cosmochimica Acta, 2010, 74(1):264-273.
DOI URL |
| [85] |
EGGENKAMP H G M, KREULEN R, VAN GROOS A F K. Chlorine stable isotope fractionation in evaporites[J]. Geochimica et Cosmochimica Acta, 1995, 59(24):5169-5175.
DOI URL |
| [86] |
EASTOE C J, LONG A, KNAUTH L P. Stable chlorine isotopes in the Palo Duro Basin, Texas: evidence for preservation of Permian evaporite brines[J]. Geochimica et Cosmochimica Acta, 1999, 63(9):1375-1382.
DOI URL |
| [87] |
EASTOE C J, PERYT T. Stable chlorine isotope evidence for non-marine chloride in Badenian evaporites, Carpathian mountain region[J]. Terra Nova, 1999, 11(2/3):118-131.
DOI URL |
| [88] | LINDEMANN F A. Discussion on isotopes[J]. Proceedings of the Royal Society of London Series A, 1921, 99:102-104. |
| [89] |
RICHTER F M, MENDYBAEV R A, CHRISTENSEN J N, et al. Kinetic isotopic fractionation during diffusion of ionic species in water[J]. Geochimica et Cosmochimica Acta, 2006, 70(2):277-289.
DOI URL |
| [90] |
BOURG I C, RICHTER F M, CHRISTENSEN J N, et al. Isotopic mass dependence of metal cation diffusion coefficients in liquid water[J]. Geochimica et Cosmochimica Acta, 2010, 74(8):2249-2256.
DOI URL |
| [91] | KONSTANTINOV B P, BAKULIN E A. Separation of chlorine isotopes in aqueous LiCl, NaCl and HCl solutions[J]. Zhurnal Fizicheskoi Khimii (USSR) (For English Translation See Russian Journal of Physical Chemistry B), 1965, 39(3):592-596. |
| [92] |
EGGENKAMP H G M, COLEMAN M L. The effect of aqueous diffusion on the fractionation of chlorine and bromine stable isotopes[J]. Geochimica et Cosmochimica Acta, 2009, 73(12):3539-3548.
DOI URL |
| [93] |
GROEN J, VELSTRA J, MEESTERS A. Salinization processes in paleowaters in coastal sediments of Suriname: evidence from δ37Cl analysis and diffusion modelling[J]. Journal of Hydrology, 2000, 234(1/2):1-20.
DOI URL |
| [94] |
BEEKMAN H E, EGGENKAMP H G M, APPELO C A J. An integrated modelling approach to reconstruct complex solute transport mechanisms: Cl and δ37Cl in pore water of sediments from a former brackish lagoon in the Netherlands[J]. Applied Geochemistry, 2011, 26(3):257-268.
DOI URL |
| [95] |
GODON A, JENDRZEJEWSKI N, CASTREC-ROUELLE M, et al. Origin and evolution of fluids from mud volcanoes in the Barbados accretionary complex[J]. Geochimica et Cosmochimica Acta, 2004, 68(9):2153-2165.
DOI URL |
| [96] |
ZIEGLER K, COLEMAN M L, HOWARTH R J. Palaeohydrodynamics of fluids in the Brent Group (Oseberg Field, Norwegian North Sea) from chemical and isotopic compositions of formation waters[J]. Applied Geochemistry, 2001, 16(6):609-632.
DOI URL |
| [97] |
SHARP Z D, MERCER J A, JONES R H, et al. The chlorine isotope composition of chondrites and Earth[J]. Geochimica et Cosmochimica Acta, 2013, 107:189-204.
DOI URL |
| [98] | BARNES J D, SHARP Z D, FISCHER T P, et al. Chlorine isotope variations along the Central American volcanic front and back arc[J]. Geochemistry, Geophysics, Geosystems, 2009, 10(11): Q11S17. |
| [99] |
BARNES J D, SHARP Z D. Chlorine isotope geochemistry[J]. Reviews in Mineralogy and Geochemistry, 2017, 82(1):345-378.
DOI URL |
| [100] | RIZZO A L, CARACAUSI A, LIOTTA M, et al. Chlorine isotope composition of volcanic gases and rocks at Mount Etna (Italy) and inferences on the local mantle source[J]. Earth and Planetary Science Letters, 2013, 371:134-142. |
| [101] |
COLEMAN M L, ADER M, CHAUDHURI S, et al. Microbial isotopic fractionation of perchlorate chlorine[J]. Applied and Environmental Microbiology, 2003, 69(8):4997-5000.
DOI URL |
| [102] | WANG Y, HSU W, GUAN Y. An extremely heavy chlorine reservoir in the Moon: insights from the apatite in lunar meteorites[J]. Scientific Reports, 2019, 9(1):1-8. |
| [103] |
FARLEY K A, MARTIN P, ARCHER P DJr, et al. Light and variable 37Cl/35Cl ratios in rocks from Gale Crater, Mars: possible signature of perchlorate[J]. Earth and Planetary Science Letters, 2016, 438:14-24.
DOI URL |
| [104] |
RANSOM B, SPIVACK A J, KASTNER M. Stable Cl isotopes in subduction-zone pore waters: implications for fluid-rock reactions and the cycling of chlorine[J]. Geology, 1995, 23(8):715-718.
DOI URL |
| [105] | KAUFMANN R S, FRAPE S K, FRITZ P, et al. Chlorine stable isotope composition of Canadian Shield brines[J]. Saline Water and Gases in Crystalline Rocks, 1987, 33:89-93. |
| [106] |
KAUFMANN R S, FRAPE S K, MCNUTT R, et al. Chlorine stable isotope distribution of Michigan Basin formation waters[J]. Applied Geochemistry, 1993, 8(4):403-407.
DOI URL |
| [107] | KAUFMANN R S, LONG A, CAMPBELL D J. Chlorine isotope distribution in formation waters, Texas and Louisiana[J]. AAPG Bulletin, 1988, 72(7):839-844. |
| [108] |
EASTOE C J, LONG A, LAND L S, et al. Stable chlorine isotopes in halite and brine from the Gulf Coast Basin: brine genesis and evolution[J]. Chemical Geology, 2001, 176(1/2/3/4):343-360.
DOI URL |
| [109] |
SHOUAKAR-STASH O, ALEXEEV S V, FRAPE S K, et al. Geochemistry and stable isotopic signatures, including chlorine and bromine isotopes, of the deep groundwaters of the Siberian Platform, Russia[J]. Applied Geochemistry, 2007, 22(3):589-605.
DOI URL |
| [110] |
STOTLER R L, FRAPE S K, SHOUAKAR-STASH O. An isotopic survey of δ81Br and δ37Cl of dissolved halides in the Canadian and Fennoscandian Shields[J]. Chemical Geology, 2010, 274(1/2):38-55.
DOI URL |
| [111] | STURCHIO N C, BÖHLKE J K, GU B, et al. Stable isotopic composition of chlorine and oxygen in synthetic and natural perchlorate[M]// Perchlorate. Boston: Springer, 2006: 93-109. |
| [112] |
JACKSON W A, BÖHLKE J K, GU B, et al. Isotopic composition and origin of indigenous natural perchlorate and co-occurring nitrate in the southwestern United States[J]. Environmental Science & Technology, 2010, 44(13):4869-4876.
DOI URL |
| [113] |
SELVERSTONE J, SHARP Z D. Chlorine isotope behavior during prograde metamorphism of sedimentary rocks[J]. Earth and Planetary Science Letters, 2015, 417:120-131.
DOI URL |
| [114] |
BARNES J D, SHARP Z D. A chlorine isotope study of DSDP/ODP serpentinized ultramafic rocks: insights into the serpentinization process[J]. Chemical Geology, 2006, 228(4):246-265.
DOI URL |
| [115] | BARNES J D, CISNEROS M. Mineralogical control on the chlorine isotope composition of altered oceanic crust[J]. Chemical Geology, 2012, 326:51-60. |
| [116] |
BARNES J D, ELDAM R, LEE C T A, et al. Petrogenesis of serpentinites from the Franciscan Complex, western California, USA[J]. Lithos, 2013, 178:143-157.
DOI URL |
| [117] |
TREIMAN A H, BOYCE J W, GROSS J, et al. Phosphate-halogen metasomatism of lunar granulite 79215: impact-induced fractionation of volatiles and incompatible elements[J]. American Mineralogist, 2014, 99(10):1860-1870.
DOI URL |
| [118] |
BOYCE J W, TREIMAN A H, GUAN Y, et al. The chlorine isotope fingerprint of the lunar magma ocean[J]. Science Advances, 2015, 1(8):e1500380.
DOI URL |
| [119] | SHARP Z, WILLIAMS J, SHERARER C, et al. The chlorine isotope composition of Martian meteorites 2: implications for the early solar system and the formation of Mars[J]. Meteoritics & Planetary Science, 2016, 51(11):2111-2126. |
| [120] |
BELLUCCI J J, WHITEHOUSE M J, JOHN T, et al. Halogen and Cl isotopic systematics in Martian phosphates: implications for the Cl cycle and surface halogen reservoirs on Mars[J]. Earth and Planetary Science Letters, 2017, 458:192-202.
DOI URL |
| [121] | MCCUBBIN F M, BOYCE J W, SRINIVASAN P, et al. Heterogeneous distribution of H2O in the Martian interior: implications for the abundance of H2O in depleted and enriched mantle sources[J]. Meteoritics & Planetary Science, 2016, 51(11):2036-2060. |
| [122] | GARGANO A M, SHARP Z D. The chlorine isotope composition of the Solar Nebula & implications to the sources of volatiles to the terrestrial planets[R]. New Orleans: American Geophysical Union Fall Meeting, 2017. |
| [123] | GARGANO A M, SHARP Z D. The chlorine isotope composition of iron meteorites: evidence for the Cl isotope composition of the solar nebula and implications for extensive devolatilization during planet formation[J]. Meteoritics & Planetary Science, 2019, 54(7):1619-1631. |
| [124] |
LUCEY P, KOROTEV R L, GILLIS J J, et al. Understanding the lunar surface and space-Moon interactions[J]. Reviews in Mineralogy and Geochemistry, 2006, 60(1):83-219.
DOI URL |
| [125] |
TAYLOR S R, PIETERS C M, MACPHERSON G J. Earth-Moon system, planetary science, and lessons learned[J]. Reviews in Mineralogy and Geochemistry, 2006, 60(1):657-704.
DOI URL |
| [126] |
WIECZOREK M A, JOLLIFF B L, KHAN A, et al. The constitution and structure of the lunar interior[J]. Reviews in Mineralogy and Geochemistry, 2006, 60(1):221-364.
DOI URL |
| [127] | EPSTEIN S, TAYLOR JR H P. 18O/16O, 30Si/28Si, D/H and 13C/12C ratios in lunar samples[C]// Lunar and planetary science conference proceedings. Texas: LPI Contribution No. 1569, 1971, 2:1421. |
| [128] | THODE H G, REES C E. Sulphur isotopes in grain size fractions of lunar soils[C]// Lunar and planetary science conference proceedings. Texas: LPI Contribution No.1573, 1976, 7:459-468. |
| [129] |
SHARP Z D, MCCUBBIN F M, SHEARER C K. A hydrogen-based oxidation mechanism relevant to planetary formation[J]. Earth and Planetary Science Letters, 2013, 380:88-97.
DOI URL |
| [130] |
CAVOSIE A J, VALLEY J W, WILDE S A. Magmatic δ18O in 4400-3900 Ma detrital zircons: a record of the alteration and recycling of crust in the Early Archean[J]. Earth and Planetary Science Letters, 2005, 235(3/4):663-681.
DOI URL |
| [131] |
BONIFACIE M, JENDRZEJEWSKI N, AGRINIER P, et al. The chlorine isotope composition of Earth's mantle[J]. Science, 2008, 319(5869):1518-1520.
DOI URL |
| [132] |
LAYNE G D, KENT A J R, BACH W. δ37Cl systematics of a backarc spreading system: the Lau Basin[J]. Geology, 2009, 37(5):427-430.
DOI URL |
| [133] |
PINTI D L, SHOUAKAR-STASH O, CASTRO M C, et al. The bromine and chlorine isotopic composition of the mantle as revealed by deep geothermal fluids[J]. Geochimica et Cosmochimica Acta, 2020, 276:14-30.
DOI URL |
| [134] | CHEN Y H. Sequence of salt separation and regularity of some trace-elements distribution during isothermal evaporation (25 ℃) of the Huanghai seawater[J]. Acta Geologica Sinica, 1983, 57(4):379-390. |
| [135] |
LUO C, XIAO Y, WEN H, et al. Stable isotope fractionation of chlorine during the precipitation of single chloride minerals[J]. Applied Geochemistry, 2014, 47:141-149.
DOI URL |
| [136] |
ARCURI T, BRIMHALL G. The chloride source for atacamite mineralization at the Radomiro Tomic porphyry copper deposit, northern Chile[J]. Economic Geology, 2003, 98(8):1667-1681.
DOI URL |
| [137] |
SHARP Z D, BARNES J D. Water-soluble chlorides in massive seafloor serpentinites: a source of chloride in subduction zones[J]. Earth and Planetary Science Letters, 2004, 226(1/2):243-254.
DOI URL |
| [138] |
LAUBE J C, KAISER J, STURGES W T, et al. Chlorine isotope fractionation in the stratosphere[J]. Science, 2010, 329(5996):1167-1167.
DOI URL |
| [139] |
BRENNINKMEIJER C A M, JANSSEN C, KAISER J, et al. Isotope effects in the chemistry of atmospheric trace compounds[J]. Chemical Reviews, 2003, 103(12):5125-5162.
DOI URL |
| [140] |
BERNAL N F, GLEESON S A, DEAN A S, et al. The source of halogens in geothermal fluids from the Taupo Volcanic Zone, North Island, New Zealand[J]. Geochimica et Cosmochimica Acta, 2014, 126:265-283.
DOI URL |
| [141] |
CULLEN J, BARNES J D, HURWITZ S, et al. Halogen and chlorine isotope composition of thermal springs along and across the Cascadia arc[J]. Earth and Planetary Science Letters, 2015, 426:225-234.
DOI URL |
| [142] |
LI L, BONIFACIE M, AUBAUD C, et al. Chlorine isotopes of thermal springs in arc volcanoes for tracing shallow magmatic activity[J]. Earth and Planetary Science Letters, 2015, 413:101-110.
DOI URL |
| [143] |
ENGLAND P, ENGDAHL R, THATCHER W. Systematic variation in the depths of slabs beneath arc volcanoes[J]. Geophysical Journal International, 2004, 156(2):377-408.
DOI URL |
| [144] |
ZHANG M, FRAPE S K, LOVE A J, et al. Chlorine stable isotope studies of old groundwater, southwestern Great Artesian Basin, Australia[J]. Applied Geochemistry, 2007, 22(3):557-574.
DOI URL |
| [145] |
NAHNYBIDA T, GLEESON S A, RUSK B G, et al. Cl/Br ratios and stable chlorine isotope analysis of magmatic-hydrothermal fluid inclusions from Butte, Montana and Bingham Canyon, Utah[J]. Mineralium Deposita, 2009, 44(8):837.
DOI URL |
| [146] |
REDMOND P B, EINAUDI M T, INAN E E, et al. Copper deposition by fluid cooling in intrusion-centered systems: new insights from the Bingham porphyry ore deposit, Utah[J]. Geology, 2004, 32(3):217-220.
DOI URL |
| [147] | WILLIAMS P J, BARTON M D, JOHNSON D A, et al. Iron oxide copper-gold deposits: geology, space-time distribution, and possible modes of origin[J]. Economic Geology, 2005: 371-405. |
| [148] |
EASTOE C J, GUILBERT J M, KAUFMANN R S. Preliminary evidence for fractionation of stable chlorine isotopes in ore-forming hydrothermal systems[J]. Geology, 1989, 17(3):285-288.
DOI URL |
| [149] | ROEDDER E. Fluid inclusions[J]. Reviews in Mineralogy and Geochemistry, 1984, 12:79-148. |
| [150] |
EASTOE C J, GUILBERT J M. Stable chlorine isotopes in hydrothermal processes[J]. Geochimica et Cosmochimica Acta, 1992, 56(12):4247-4255.
DOI URL |
| [151] |
EGGENKAMP H G M, SCHUILING R D. δ37Cl variations in selected minerals: a possible tool for exploration[J]. Journal of Geochemical Exploration, 1995, 55(1/2/3):249-255.
DOI URL |
| [152] |
STEWART M A, SPIVACK A J. The stable-chlorine isotope compositions of natural and anthropogenic materials[J]. Reviews in Mineralogy and Geochemistry, 2004, 55(1):231-254.
DOI URL |
| [153] |
GLEESON S A, SMITH M P. The sources and evolution of mineralising fluids in iron oxide-copper-gold systems, Norrbotten, Sweden: constraints from Br/Cl ratios and stable Cl isotopes of fluid inclusion leachates[J]. Geochimica et Cosmochimica Acta, 2009, 73(19):5658-5672.
DOI URL |
| [154] |
CHIARADIA M, BANKS D, CLIFF R, et al. Origin of fluids in iron oxide-copper-gold deposits: constraints from δ37Cl, 87Sr/86Sri and Cl/Br[J]. Mineralium Deposita, 2006, 41(6):565-573.
DOI URL |
| [155] |
HANLEY J, AMES D, BARNES J, et al. Interaction of magmatic fluids and silicate melt residues with saline groundwater in the footwall of the Sudbury Igneous Complex, Ontario, Canada: new evidence from bulk rock geochemistry, fluid inclusions and stable isotopes[J]. Chemical Geology, 2011, 281(1/2):1-25.
DOI URL |
| [156] |
ANNABLE W K, FRAPE S K, SHOUAKAR-STASH O, et al. 37Cl, 15N, 13C isotopic analysis of common agro-chemicals for identifying non-point source agricultural contaminants[J]. Applied Geochemistry, 2007, 22(7):1530-1536.
DOI URL |
| [157] |
SHOUAKAR-STASH O, FRAPE S K, DRIMMIE R J. Stable hydrogen, carbon and chlorine isotope measurements of selected chlorinated organic solvents[J]. Journal of Contaminant Hydrology, 2003, 60(3/4):211-228.
DOI URL |
| [158] |
JENDRZEJEWSKI N, EGGENKAMP H G M, COLEMAN M L. Characterisation of chlorinated hydrocarbons from chlorine and carbon isotopic compositions: scope of application to environmental problems[J]. Applied Geochemistry, 2001, 16(9/10):1021-1031.
DOI URL |
| [159] |
CHEN G, SHOUAKAR-STASH O, PHILLIPS E, et al. Dual carbon-chlorine isotope analysis indicates distinct anaerobic dichloromethane degradation pathways in two members of Peptococcaceae[J]. Environmental Science & Technology, 2018, 52(15):8607-8616.
DOI URL |
| [160] |
FILIPPELLI G M. The global phosphorus cycle: past, present, and future[J]. Elements, 2008, 4(2):89-95.
DOI URL |
| [161] |
FILIPPELLI G M. The global phosphorus cycle[J]. Reviews in Mineralogy and Geochemistry, 2002, 48(1):391-425.
DOI URL |
| [162] |
SOMMERAUER J, KATZ-LEHNERT K. A new partial substitution mechanism of $CO_{3}^{2-}$/CO3OH3- and $SiO_{4}^{4-}$ for the $PO_4^{3-}$ group in hydroxyapatite from the Kaiserstuhl alkaline complex (SW-Germany)[J]. Contributions to Mineralogy and Petrology, 1985, 91(4):360-368.
DOI URL |
| [163] |
PAN Y, FLEET M E. Compositions of the apatite-group minerals: substitution mechanisms and controlling factors[J]. Reviews in Mineralogy and Geochemistry, 2002, 48(1):13-49.
DOI URL |
| [164] |
HUGHES J M, RAKOVAN J F. Structurally robust, chemically diverse: apatite and apatite supergroup minerals[J]. Elements, 2015, 11(3):165-170.
DOI URL |
| [165] | HUGHES J M, CAMERON M, CROWLEY K D. Structural variations in natural F, OH, and Cl apatites[J]. American Mineralogist, 1989, 74(7/8):870-876. |
| [166] |
HUGHES J M, RAKOVAN J. The crystal structure of apatite, Ca5(PO4)3(F, OH, Cl)[J]. Reviews in Mineralogy and Geochemistry, 2002, 48(1):1-12.
DOI URL |
| [167] |
ZHU C, SVERJENSKY D A. Partitioning of F-Cl-OH between minerals and hydrothermal fluids[J]. Geochimica et Cosmochimica Acta, 1991, 55(7):1837-1858.
DOI URL |
| [168] | O’REILLY S Y, GRIFFIN W L. Mantle metasomatism[M]// Metasomatism and the chemical transformation of rock. Berlin: Springer, 2013: 471-533. |
| [169] | ELLIOTT J C. Structure and chemistry of the apatites and other calcium orthophosphates[M]. Amsterdam: Elsevier, 2013. |
| [170] | KOHN M J, RAKOVAN J, HUGHES J M. Phosphates: geochemical, geobiological, and materials importance[J]. Reviews in Mineralogy & Geochemistry, 2002, 48:294-335. |
| [171] | BOUDREAU A E, MCCALLUM I S. Low temperature alteration of REE-rich chlorapatite from the Stillwater Complex, Montana[J]. American Mineralogist, 1990, 75(5/6):687-693. |
| [172] |
HARLOV D E. Apatite: a fingerprint for metasomatic processes[J]. Elements, 2015, 11(3):171-176.
DOI URL |
| [173] |
ANDERSSON S S, WAGNER T, JONSSON E, et al. Apatite as a tracer of the source, chemistry and evolution of ore-forming fluids: the case of the Olserum-Djupedal REE-phosphate mineralisation, SE Sweden[J]. Geochimica et Cosmochimica Acta, 2019, 255:163-187.
DOI URL |
| [174] |
O’REILLY S Y, GRIFFIN W L. Apatite in the mantle: implications for metasomatic processes and high heat production in Phanerozoic mantle[J]. Lithos, 2000, 53(3/4):217-232.
DOI URL |
| [175] |
BOYCE J W, HERVIG R L. Apatite as a monitor of late-stage magmatic processes at Volcán Irazú, Costa Rica[J]. Contributions to Mineralogy and Petrology, 2009, 157(2):135.
DOI URL |
| [176] |
FARLEY K A, SHUSTER D L, KETCHAM R A. U and Th zonation in apatite observed by laser ablation ICPMS, and implications for the (U-Th)/He system[J]. Geochimica et Cosmochimica Acta, 2011, 75(16):4515-4530.
DOI URL |
| [177] |
WILLIAMS M L, JERCINOVIC M J, HETHERINGTON C J. Microprobe monazite geochronology: understanding geologic processes by integrating composition and chronology[J]. Annual Review of Earth and Planetary Sciences, 2007, 35:137-175.
DOI URL |
| [178] |
BOYCE J W, TOMLINSON S M, MCCUBBIN F M, et al. The lunar apatite paradox[J]. Science, 2014, 344(6182):400-402.
DOI URL |
| [179] |
MCCUBBIN F M, STEELE A, HAURI E H, et al. Nominally hydrous magmatism on the Moon[J]. Proceedings of the National Academy of Sciences, 2010, 107(25):11223-11228.
DOI URL |
| [180] |
BOYCE J W, LIU Y, ROSSMAN G R, et al. Lunar apatite with terrestrial volatile abundances[J]. Nature, 2010, 466(7305):466-469.
DOI URL |
| [181] |
BARNES J J, TARTESE R, ANAND M, et al. Early degassing of lunar urKREEP by crust-breaching impact(s)[J]. Earth and Planetary Science Letters, 2016, 447:84-94.
DOI URL |
| [182] |
BOYCE J W, KANEE S A, MCCUBBIN F M, et al. Early loss, fractionation, and redistribution of chlorine in the Moon as revealed by the low-Ti lunar mare basalt suite[J]. Earth and Planetary Science Letters, 2018, 500:205-214.
DOI URL |
| [183] |
STEPHANT A, ANAND M, ZHAO X, et al. The chlorine isotopic composition of the Moon: insights from melt inclusions[J]. Earth and Planetary Science Letters, 2019, 523:115715.
DOI URL |
| [1] | 刘金萍, 王改云, 简晓玲, 朱传庆, 胡小强, 袁晓蔷, 王超. 北黄海东部次盆地构造热机制与成烃效应[J]. 地学前缘, 2024, 31(4): 206-218. |
| [2] | 李茜, 朱光有, 李婷婷, 陈志勇, 艾依飞, 张岩, 田连杰. U同位素分馏行为及其在环境地球科学中的应用研究进展[J]. 地学前缘, 2024, 31(2): 447-471. |
| [3] | 聂潇, 陈雷, 郭现轻, 于涛, 王宗起. 南秦岭中段宁陕地区绿柱石-铌铁矿型伟晶岩中磷灰石和铌铁矿族矿物的矿物地球化学研究[J]. 地学前缘, 2023, 30(5): 115-133. |
| [4] | 刘嘉文, 田世洪, 王玲. 镁同位素体系在重要地质过程中的应用[J]. 地学前缘, 2023, 30(3): 399-424. |
| [5] | 张卫民, 王振, 钱程, 郭亚丹, 刘海燕. 方解石负载羟基磷灰石复合材料去除水中铀离子的PRB活性介质研究[J]. 地学前缘, 2021, 28(5): 175-185. |
| [6] | 李源, 王长秋, 鲁安怀, 李艳, 杨重庆, 李康. 脑动脉粥样硬化斑块中不同类型钙化集合体的矿物学特征及分布规律[J]. 地学前缘, 2020, 27(5): 291-299. |
| [7] | 骆少勇, 周跃飞, 刘星. 磷灰石对湖泊沉积物中水铁矿稳定性的制约[J]. 地学前缘, 2020, 27(5): 218-226. |
| [8] | 刘茜, 王奕菁, 魏海珍. 稳定氯同位素地球化学研究进展[J]. 地学前缘, 2020, 27(3): 29-41. |
| [9] | 赵新福, 曾丽平, 廖旺, 李婉婷, 胡浩, 李建威. 长江中下游成矿带玢岩铁矿研究新进展及对矿床成因的启示[J]. 地学前缘, 2020, 27(2): 197-217. |
| [10] | 陈雪,袁万明,袁二军,王珂,冯子睿. 青海东昆仑东山根矿区构造活动的磷灰石裂变径迹分析[J]. 地学前缘, 2018, 25(6): 330-337. |
| [11] | 朱传庆,邱楠生,曹环宇,刘一锋,江强. 四川盆地东部构造热演化:来自镜质体反射率和磷灰石裂变径迹的约束[J]. 地学前缘, 2017, 24(3): 94-104. |
| [12] | 常健,邱楠生. 磷灰石低温热年代学技术及在塔里木盆地演化研究中的应用[J]. 地学前缘, 2017, 24(3): 79-93. |
| [13] | 朱建明, 谭德灿, 王静, 曾理. 硒同位素地球化学研究进展与应用[J]. 地学前缘, 2015, 22(5): 102-114. |
| [14] | 王泽洲, 刘盛遨, 李丹丹, 吕逸文, 吴松, 赵云. 铜同位素地球化学及研究新进展[J]. 地学前缘, 2015, 22(5): 72-83. |
| [15] | 祝红丽, 张兆峰, 刘峪菲, 刘芳, 康晋霆. 钙同位素地球化学综述[J]. 地学前缘, 2015, 22(5): 44-53. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
