

地学前缘 ›› 2021, Vol. 28 ›› Issue (5): 68-78.DOI: 10.13745/j.esf.sf.2021.2.19
惠淑君1( ), 杨冰1,2, 郭华明1,*(
), 杨冰1,2, 郭华明1,*( ), 连国玺2, 孙娟2
), 连国玺2, 孙娟2
收稿日期:2020-08-17
修回日期:2021-01-15
出版日期:2021-09-25
发布日期:2021-10-29
通讯作者:
郭华明
作者简介:惠淑君(1994—),女,硕士,主要从事环境科学与工程相关研究。E-mail: 1365235784@qq.com
基金资助:
HUI Shujun1( ), YANG Bing1,2, GUO Huaming1,*(
), YANG Bing1,2, GUO Huaming1,*( ), LIAN Guoxi2, SUN Juan2
), LIAN Guoxi2, SUN Juan2
Received:2020-08-17
Revised:2021-01-15
Online:2021-09-25
Published:2021-10-29
Contact:
GUO Huaming
摘要:
铀矿开采过程中及井场退役后,含铀浸出液的扩散会对地下水造成一定影响,该影响范围和程度决定了铀的天然衰减特征。本文以北方某地浸铀矿区砂岩含水介质为研究对象,研究了溶液化学特征和黄铁矿含量对砂岩吸附和转化铀的影响。结果表明,砂岩颗粒对U(VI)的吸附基本在12 h可达平衡。线性等温吸附模型可以很好地描述吸附特征;砂岩颗粒对U(VI)的吸附率总体随着粒径增大而减小,当岩石粒径增大到0.200.25 mm时,吸附率趋于稳定。溶液pH值是影响吸附的主要因素,通过控制溶液中U(VI)的络合形态和岩石颗粒表面带电荷情况,在库仑力的作用下促进或者抑制吸附,在pH=6时,达到最佳吸附状态。共存离子对U(VI)吸附的抑制程度是:
中图分类号:
惠淑君, 杨冰, 郭华明, 连国玺, 孙娟. 不同因素对砂岩含水层介质吸附铀的影响[J]. 地学前缘, 2021, 28(5): 68-78.
HUI Shujun, YANG Bing, GUO Huaming, LIAN Guoxi, SUN Juan. Factors affecting uranium adsorption on aquifer sandstone[J]. Earth Science Frontiers, 2021, 28(5): 68-78.
| 粒径/mm | R2 | Kd/(L·g-1) |
|---|---|---|
| <0.10 | 0.995 1 | 0.030 |
| 0.10<0.15 | 0.994 3 | 0.038 |
| 0.15<0.20 | 0.997 9 | 0.032 |
| 0.20<0.25 | 0.993 6 | 0.029 |
| 0.250.50 | 0.997 8 | 0.029 |
表1 不同粒径砂岩样品等温吸附曲线拟合系数和吸附分配系数
Table 1 Fitting coefficients and adsorption partition coefficients of isothermal adsorption curves for sandstone samples with different particle sizes
| 粒径/mm | R2 | Kd/(L·g-1) |
|---|---|---|
| <0.10 | 0.995 1 | 0.030 |
| 0.10<0.15 | 0.994 3 | 0.038 |
| 0.15<0.20 | 0.997 9 | 0.032 |
| 0.20<0.25 | 0.993 6 | 0.029 |
| 0.250.50 | 0.997 8 | 0.029 |
| 因素 | 不同pH值条件下的影响因素情况 | ||
|---|---|---|---|
| pH<4 | pH=46 | pH>6 | |
| 固体表面带电荷情况 | 有正电荷也有负电荷 | 主要带负电荷 | 几乎都带负电荷 |
| U的络合形态 | | (UO2)3(OH (UO2)4(OH | UO2CO3、UO2(OH (UO2)3(OH UO2(CO3 |
| 库仑力 | 引力为主 | 引力 | 斥力增强 |
| H+竞争作用 | 较强 | 减弱 | 几乎没有 |
表2 不同pH值条件下影响U(VI)吸附的因素
Table 2 List of factors affecting U(VI) adsorption at different pH
| 因素 | 不同pH值条件下的影响因素情况 | ||
|---|---|---|---|
| pH<4 | pH=46 | pH>6 | |
| 固体表面带电荷情况 | 有正电荷也有负电荷 | 主要带负电荷 | 几乎都带负电荷 |
| U的络合形态 | | (UO2)3(OH (UO2)4(OH | UO2CO3、UO2(OH (UO2)3(OH UO2(CO3 |
| 库仑力 | 引力为主 | 引力 | 斥力增强 |
| H+竞争作用 | 较强 | 减弱 | 几乎没有 |
| 不同试验条件 | U(VI)含量占比/% | U(IV)含量占比/% | Fe(II)含量占比/% | Fe(III)含量占比/% |
|---|---|---|---|---|
| 原始黄铁矿 | 98.9 | 1.1 | ||
| 近中性pH值 | 68 | 32 | 95.8 | 4.2 |
| 弱碱性pH值 | 100 | 0 | 98.5 | 1.5 |
表3 不同试验条件下10% FeS2组反应10 d的固体表面U(VI)、U(IV)及Fe(II)、Fe(III)含量占比
Table 3 Percentages of U (VI), U (IV), Fe(II), and Fe(III) on solid sample surface containing 10% FeS2 reacting with U(VI) for 10 d under different experimental conditions
| 不同试验条件 | U(VI)含量占比/% | U(IV)含量占比/% | Fe(II)含量占比/% | Fe(III)含量占比/% |
|---|---|---|---|---|
| 原始黄铁矿 | 98.9 | 1.1 | ||
| 近中性pH值 | 68 | 32 | 95.8 | 4.2 |
| 弱碱性pH值 | 100 | 0 | 98.5 | 1.5 |
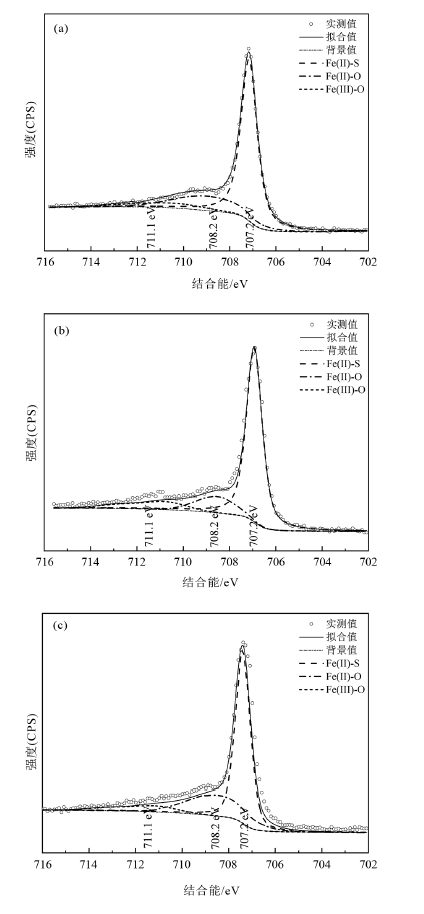
图10 不同试验条件下10% FeS2组反应10 d的固体Fe(2p)XPS光谱 a—原始黄铁矿;b—近中性pH值;c—弱碱性pH值。
Fig.10 Iron 2p XPS spectra for solid samples containing 10% FeS2 reacting with U(VI) for 10 d under different experimental conditions
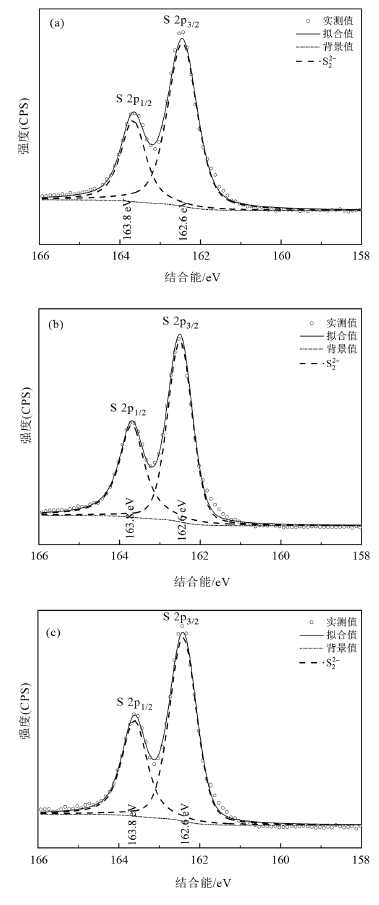
图11 不同试验条件下10% FeS2组反应10 d的固体S(2p)XPS光谱 a—原始黄铁矿;b—近中性pH值;c—弱碱性pH值。
Fig.11 Sulphur 2p XPS spectra for solid samples containing 10% FeS2 reacting with U(VI) for 10 d under different experimental conditions
| [1] | 张飞凤, 苏学斌, 邢拥国, 等. 地浸采铀新工艺综述[J]. 中国矿业, 2012, 21(增刊):9-12. |
| [2] |
SEREDKIN M, ZABOLOTSKY A, JEFFRESS G. In situ recovery, an alternative to conventional methods of mining: exploration, resource estimation, environmental issues, project evaluation and economics[J]. Ore Geology Reviews, 2016, 79:500-514.
DOI URL |
| [3] | 苏学斌. 高效绿色发展推进铀矿大基地建设[J]. 中国核工业, 2016(11):16-19. |
| [4] | 李衡, 周义朋. 地浸采铀溶质运移研究进展及展望[J]. 稀有金属, 2019, 43(3):319-330. |
| [5] | International Atomic Energy Agency. In situ leach uranium mining: an overview of operations[R]. Vienna: IAEA, 2016. |
| [6] |
CORLIN L, ROCK T, CORDOVA J, et al. Health effects and environmental justice concerns of exposure to uranium in drinking water[J]. Current Environmental Health Reports, 2016, 3(4):434-442.
DOI URL |
| [7] |
DICKINSON M, SCOTT T B. The application of zero-valent iron nanoparticles for the remediation of a uranium-contaminated waste effluent[J]. Journal of Hazardous Materials, 2010, 178(1/2/3):171-179.
DOI URL |
| [8] |
BORCH T, ROCHE N, JOHNSON T E. Determination of contaminant levels and remediation efficacy in groundwater at a former in situ recovery uranium mine[J]. Journal of Environmental Monitoring, 2012, 14(7):1814-1823.
DOI URL |
| [9] |
STANLEY D M, WILKIN R T. Solution equilibria of uranyl minerals:role of the common groundwater ions calcium and carbonate[J]. Journal of Hazardous Materials, 2019, 377:315-320.
DOI URL |
| [10] |
BACHMAF S, PLANER-FRIEDRICH B, MERKEL B J. Effect of sulfate, carbonate, and phosphate on the uranium(VI) sorption behavior onto bentonite[J]. Radiochimica Acta, 2008, 96(6):359-366.
DOI URL |
| [11] | GAJOWIAK A, MAJDAN M, DROZDZAL K. Sorption of uranium(VI) on clays and clay minerals[J]. Przemysl Chemiczny, 2009, 88(2):190-196. |
| [12] |
WEI M, LIAO J L, LIU N, et al. Interaction between uranium and humic acid (I):adsorption behaviors of U(VI) in soil humic acids[J]. Nuclear Science and Techniques, 2007, 18(5):287-293.
DOI URL |
| [13] |
XIE S B, ZHANG C, ZHOU X H, et al. Removal of uranium (VI) from aqueous solution by adsorption of hematite[J]. Journal of Environmental Radioactivity, 2009, 100(2):162-166.
DOI URL |
| [14] |
AL-HOBAIB A S, AL-SUHYBANI A A. Removal of uranyl ions from aqueous solutions using barium titanate[J]. Journal of Radioanalytical and Nuclear Chemistry, 2014, 299(1):559-567.
DOI URL |
| [15] | 吴晓朦, 刘洪雪, 王锐, 等. 硝酸铀酰溶液初始浓度与pH值对其水解反应的影响[J]. 辽宁石油化工大学学报, 2015, 35(5):18-21. |
| [16] |
FORNASIERO D, EIJT V, RALSTON J. An electrokinetic study of pyrite oxidation[J]. Colloids and Surfaces, 1992, 62(1/2):63-73.
DOI URL |
| [17] |
BEBIE J, SCHOONEN M A A, FUHRMANN M, et al. Surface charge development on transition metal sulfides:an electrokinetic study[J]. Geochimica et Cosmochimica Acta, 1998, 62(4):633-642.
DOI URL |
| [18] |
LUO M B, LIU S J, LI J Q, et al. Uranium sorption characteristics onto synthesized pyrite[J]. Journal of Radioanalytical and Nuclear Chemistry, 2016, 307(1):305-312.
DOI URL |
| [19] |
NOUBACTEP C, SONNEFELD J, MERTEN D, et al. Effects of the presence of pyrite and carbonate minerals on the kinetics of the uranium release from a natural rock[J]. Journal of Radioanalytical and Nuclear Chemistry, 2006, 270(2):325-333.
DOI URL |
| [20] |
LIU J, ZHAO C S, YUAN G Y, et al. Adsorption of U(VI) on a chitosan/polyaniline composite in the presence of Ca/Mg-U(VI)-CO3 complexes[J]. Hydrometallurgy, 2018, 175:300-311.
DOI URL |
| [21] |
AUBRIET H, HUMBERT B, PERDICAKIS M. Interaction of U(VI) with pyrite, galena and their mixtures:a theoretical and multitechnique approach[J]. Radiochimica Acta, 2006, 94(9/10/11):657-663.
DOI URL |
| [22] |
EGLIZAUD N, MISERQUE F, SIMONI E, et al. Uranium(VI) interaction with pyrite (FeS2): chemical and spectroscopic studies[J]. Radiochimica Acta, 2006, 94(9/10/11):651-656.
DOI URL |
| [23] |
SCOTT T B, RIBA TORT O, ALLEN G C. Aqueous uptake of uranium onto pyrite surfaces: reactivity of fresh versus weathered material[J]. Geochimica et Cosmochimica Acta, 2007, 71(21):5044-5053.
DOI URL |
| [24] |
WUNSCH A, NAVARRE-SITCHLER A K, MOORE J, et al. Metal release from limestones at high partial-pressures of CO2[J]. Chemical Geology, 2014, 363:40-55.
DOI URL |
| [25] |
RIEGEL M. Sorption of natural uranium on weakly basic anion exchangers[J]. Solvent Extraction and Ion Exchange, 2017, 35(5):363-375.
DOI URL |
| [26] |
WU Y, WANG Y X, GUO W. Behavior and fate of geogenic uranium in a shallow groundwater system[J]. Journal of Contaminant Hydrology, 2019, 222:41-55.
DOI URL |
| [27] | 张红霞. U(Ⅵ)、Th(Ⅳ)在几种吸附剂上的吸附机理研究[D]. 兰州: 兰州大学, 2011. |
| [28] |
LI X L, WU J J, LIAO J L, et al. Adsorption and desorption of uranium (VI) in aerated zone soil[J]. Journal of Environmental Radioactivity, 2013, 115:143-150.
DOI URL |
| [29] | GUO Z, SU H Y, WU W S. Sorption and desorption of uranium(VI) on silica:experimental and modeling studies[J]. Radiochimica Acta, 2009, 97(3):133-140. |
| [30] |
BARGAR J R, REITMEYER R, DAVIS J A. Spectroscopic confirmation of uranium(VI)- carbonato adsorption complexes on hematite[J]. Environmental Science & Technology, 1999, 33(14):2481-2484.
DOI URL |
| [31] |
YANG Z W, KANG M L, MA B, et al. Inhibition of U(VI) reduction by synthetic and natural pyrite[J]. Environmental Science & Technology, 2014, 48(18):10716-10724.
DOI URL |
| [32] |
SEDER-COLOMINA M, MANGERET A, STETTEN L, et al. Carbonate facilitated mobilization of uranium from lacustrine sediments under anoxic conditions[J]. Environmental Science & Technology, 2018, 52(17):9615-9624.
DOI URL |
| [33] |
DESCOSTES M, SCHLEGEL M L, EGLIZAUD N, et al. Uptake of uranium and trace elements in pyrite (FeS2) suspensions[J]. Geochimica et Cosmochimica Acta, 2010, 74(5):1551-1562.
DOI URL |
| [34] |
LIGER E, CHARLET L, VAN CAPPELLEN P. Surface catalysis of uranium(VI) reduction by iron(II)[J]. Geochimica et Cosmochimica Acta, 1999, 63(19/20):2939-2955.
DOI URL |
| [35] |
JEON B H, DEMPSEY B A, BURGOS W D, et al. Chemical reduction of U(VI) by Fe(II) at the solid-water interface using natural and synthetic Fe(III) oxides[J]. Environmental Science & Technology, 2005, 39(15):5642-5649.
DOI URL |
| [36] | 李肃宁, 周丽, 李和平, 等. 黄铁矿吸附-还原金络合物的试验研究进展[J]. 地球与环境, 2013, 41(2):185-192. |
| [37] |
WIDLER A M, SEWARD T M. The adsorption of gold(I) hydrosulphide complexes by iron sulphide surfaces[J]. Geochimica et Cosmochimica Acta, 2002, 66(3):383-402.
DOI URL |
| [38] | CUI D Q, SPAHIU K. On the Interaction between uranyl carbonate and UO2(s) in anaerobic solution[J]. Journal of Nuclear Science and Technology, 2002, 39(Suppl 3):500-503. |
| [39] |
BRUGGEMAN C, MAES N. Uptake of uranium(VI) by pyrite under boom clay conditions:influence of dissolved organic carbon[J]. Environmental Science & Technology, 2010, 44(11):4210-4216.
DOI URL |
| [1] | 董宏坤, 万世明, 刘畅, 赵德博, 曾志刚, 李安春. 南海北部晚中新世红绿韵律层成因的矿物学和地球化学约束[J]. 地学前缘, 2022, 29(4): 42-54. |
| [2] | 崔迪, 杨冰, 郭华明, 连国玺, 孙娟. 砂岩含水介质中铀的吸附和迁移行为研究[J]. 地学前缘, 2022, 29(3): 217-226. |
| [3] | 郭华明, 高志鹏, 修伟. 地下水典型氧化还原敏感组分迁移转化的研究热点和趋势[J]. 地学前缘, 2022, 29(3): 64-75. |
| [4] | 徐林刚, 付雪瑞, 叶会寿, 郑伟, 陈勃, 方正龙. 南秦岭地区下寒武统黑色页岩赋存的千家坪大型钒矿地球化学特征及成矿环境[J]. 地学前缘, 2022, 29(1): 160-175. |
| [5] | 梁晓亮, 谭伟, 马灵涯, 朱建喜, 何宏平. 离子吸附型稀土矿床形成的矿物表/界面反应机制[J]. 地学前缘, 2022, 29(1): 29-41. |
| [6] | 黄冉笑, 王果胜, 袁国礼, 邱坤峰, Hounkpe Jechonias BIDOSSESSI. 伟晶质岩浆的同化混染与分离结晶(AFC)作用及铀成矿效应:以纳米比亚湖山铀矿为例[J]. 地学前缘, 2022, 29(1): 377-402. |
| [7] | 高珩, 谭行, 任宇, 朱乐诚, 毕二平. 土壤含水层处理系统对再生水入渗过程中“三氮”去除的柱试验模拟[J]. 地学前缘, 2021, 28(5): 125-135. |
| [8] | 张玉玲, 尹斯琦, 司超群, 王茜, 初文磊. 火山渣吸附地下水中大肠杆菌噬菌体特性研究[J]. 地学前缘, 2021, 28(5): 167-174. |
| [9] | 张卫民, 王振, 钱程, 郭亚丹, 刘海燕. 方解石负载羟基磷灰石复合材料去除水中铀离子的PRB活性介质研究[J]. 地学前缘, 2021, 28(5): 175-185. |
| [10] | 孙占学, 马文洁, 刘亚洁, 刘金辉, 周义朋. 地浸采铀矿山地下水环境修复研究进展[J]. 地学前缘, 2021, 28(5): 215-225. |
| [11] | 邵雪维, 彭永明, 王功文, 赵显勇, 唐佳洋, 黄蕾蕾, 刘晓宁, 赵宪东. 短波红外光谱、X 射线荧光光谱、黄铁矿热电性分析在胶东新城金矿田深部找矿中的应用[J]. 地学前缘, 2021, 28(3): 236-251. |
| [12] | 王彤, 朱筱敏, 董艳蕾, 杨道庆, 苏彬, 谈明轩, 刘宇, 伍炜, 张亚雄. 陆相坳陷湖盆沉积对深时古气候的响应信号: 以准噶尔盆地西北缘安集海河组为例[J]. 地学前缘, 2021, 28(1): 60-76. |
| [13] | 李成禄, 李胜荣, 袁茂文, 杜兵盈, 李文龙, Masroor ALAM, 刘东园, 刘浩. 黑龙江省嫩江—黑河构造混杂岩带科洛金矿床成因:来自黄铁矿化学成分及He-Ar、S、Pb同位素证据[J]. 地学前缘, 2020, 27(5): 99-115. |
| [14] | 骆少勇, 周跃飞, 刘星. 磷灰石对湖泊沉积物中水铁矿稳定性的制约[J]. 地学前缘, 2020, 27(5): 218-226. |
| [15] | Jinru Lin, Ning Chen, Yuanming Pan. 砷在石膏中固定机制:掺杂态和表面吸附沉淀态以及其在砷污染控制中的作用[J]. 地学前缘, 2020, 27(5): 227-237. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||